| In vivo: |
| Biosci Biotechnol Biochem. 2004 Jan;68(1):85-90. | | Triterpene acids from the leaves of Perilla frutescens and their anti-inflammatory and antitumor-promoting effects.[Pubmed: 14745168] |
METHODS AND RESULTS:
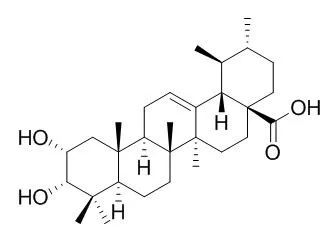
Nine triterpene acids, viz., six of the ursane type, ursolic acid (1), corosolic acid (2), 3-Epicorosolic acid (3), pomolic acid (4), tormentic acid (5) and hyptadienic acid (6), and three of the oleanane type, oleanolic acid (7), augustic acid (8) and 3-epimaslinic acid (9), among which 1 constituted the most predominant triterpene acid, were isolated and identified from ethanol extracts of the leaves of red perilla [Perilla frutescens (L.) Britton var. acuta Kudo] and green perilla [P. frutescens (L.) Britton var. acuta Kudo forma viridis Makino]. These eight compounds, 1, 2, 4-9, were evaluated for their inhibitory effects on 12-O-tetradecanoylphorbol-13-acetate (TPA)-induced inflammation (1 microg/ear) in mice.
CONCLUSIONS:
All the compounds tested showed a marked anti-inflammatory effect, with a 50% inhibitory dose (ID50) of 0.09-0.3 mg per ear. In addition, an evaluation against the Epstein-Barr virus early antigen (EBV-EA) activation induced by TPA showed five compounds, 1-3, 5 and 9, with a potent inhibitory effect on EBV-EA induction (91-93% inhibition at 1x10(3) mol ratio/TPA). Furthermore, compound 5 exhibited strong antitumor-promoting activity in an in vivo two-stage carcinogenesis test of mouse tumor by using 7,12-dimethylbenz(a)anthracene (DMBA) as an initiator and TPA as a promoter. | | Bioorg Med Chem Lett. 2015 Oct 1;25(19):4342-6. | | Synthesis of oxygenated oleanolic and ursolic acid derivatives with anti-inflammatory properties.[Pubmed: 26259803] |
METHODS AND RESULTS:
The scalable syntheses of four oxygenated triterpenes have been implemented to access substantial quantities of maslinic acid, 3-epimaslinic acid, corosolic acid, and 3-Epicorosolic acid. Semi-syntheses proceed starting from the natural products oleanolic acid and ursolic acid. Proceeding over five steps, each of the four compounds can be synthesized on the gram scale. Divergent diastereoselective reductions of α-hydroxy ketones provided access to the four targeted diol containing compounds from two precursors of the oleanane or ursane lineage. These compounds were subsequently evaluated for their ability to inhibit inflammatory gene expression in a mouse model of chemically induced skin inflammation.
CONCLUSIONS:
All compounds possessed the ability to inhibit the expression of one or more inflammatory genes induced by 12-O-tetradecanoylphorbol-13 acetate in mouse skin, however, three of the compounds, corosolic acid, 3-Epicorosolic acid and maslinic acid were more effective than the others. The availability of gram quantities will allow further testing of these compounds for potential anti-inflammatory activities as well as cancer chemopreventive activity. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)