METHODS AND RESULTS:
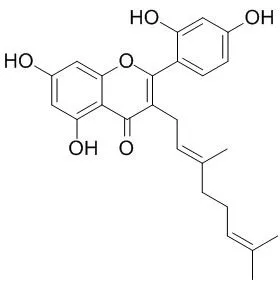
Bioassay-guided fractionation of an ethyl acetate-soluble extract from the whole plants of Broussonetia papyrifera, using an in vitro aromatase inhibition assay, led to the isolation of five new active compounds, 5,7,2',4'-Tetrahydroxy-3-geranylflavone (1), isogemichalcone C (8), 3'-[gamma-hydroxymethyl-(E)-gamma-methylallyl]-2,4,2',4'-tetrahydroxychalcone 11'-O-coumarate (9), demethylmoracin I (10), and (2S)-2',4'-dihydroxy-2' '-(1-hydroxy-1-methylethyl)dihydrofuro[2,3-h]flavanone (11), and 10 known (12-21) compounds which were also found to be active. Of these compounds, the most potent were 9 (IC(50) 0.5 microM), 11 (IC(50) 0.1 microM), isolicoflavonol (12, IC(50) 0.1 microM), and (2S)-abyssinone II (13, IC(50) 0.4 microM). Additionally, six new compounds, 5,7,3',4'-tetrahydroxy-6-geranylflavonol (2), 5,7,3',4'-tetrahydroxy-3-methoxy-6-geranylflavone (3), (2S)-7,4'-dihydroxy-3'-prenylflavan (4), 1-(2,4-dihydroxyphenyl)-3-(4-hydroxyphenyl)propane (5), 1-(2,4-dihydroxy-3-prenylphenyl)-3-(4-hydroxyphenyl)propane (6), and 1-(4-hydroxy-2-methoxyphenyl)-3-(4-hydroxy-3-prenylphenyl)propane (7), were isolated and characterized, but proved to be inactive as aromatase inhibitors, as were an additional 21 known compounds.
CONCLUSIONS:
The structures of the new compounds (1-11) were elucidated by spectroscopic methods. Structure-activity relationships in the aromatase assay were determined for the benzofurans, biphenylpropanoids, coumarins, and various types of flavonoids (chalcones, flavans, flavanones, and flavones) obtained among a total of 42 constituents of B. papyrifera. |





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)