By bioactivity-directed fractionation, five cytotoxic constituents have been characterized from the roots of Eurycoma longifolia collected in Kalimantan, Indonesia.
METHODS AND RESULTS:
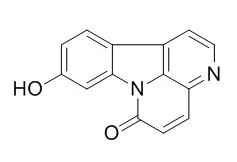
Four canthin-6-one alkaloids, namely, 9-methoxycanthin-6-one, 9-methoxycanthin-6-one-N-oxide, 9-Hydroxycanthin-6-one, and 9-Hydroxycanthin-6-one-N-oxide, and one quassinoid, eurycomanone, were found to be cytotoxic principles. Each of these compounds was evaluated against a panel of cell lines comprising a number of human cancer cell types [breast, colon, fibrosarcoma, lung, melanoma, KB, and KB-V1 (a multi-drug resistant cell line derived from KB)] and murine lymphocytic leukemia (P-388).
CONCLUSIONS:
The canthin-6-ones 1-4 were found to be active with all cell lines tested except for the KB-V1 cell line. Eurycomanone was inactive against murine lymphocytic leukemia (P-388) but was significantly active against the human cell lines tested. Two additional isolates, the beta-carboline alkaloids beta-carboline-1-propionic acid and 7-methoxy-beta-carboline-1-propionic acid, were not significantly active with these cultured cells. However, compounds 5 and 7 were found to demonstrate significant antimalarial activity as judged by studies conducted with cultured Plasmodium falciparum strains. The structures of the novel compounds 2-4 and 7 were established by spectral and chemical methods. |





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)