Erythritol is an important nutrient for several α-2 Proteobacteria, including N2-fixing plant endosymbionts and Brucella, a worldwide pathogen that finds this four-carbon polyol in genital tissues.
METHODS AND RESULTS:
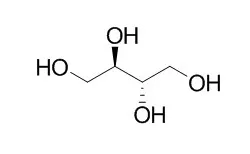
Erythritol metabolism involves phosphorylation to L-Erythritol-4-phosphate by the kinase EryA and oxidation of the latter to L-3-tetrulose 4-phosphate by the dehydrogenase EryB. It is accepted that further steps involve oxidation by the putative dehydrogenase EryC and subsequent decarboxylation to yield triose-phosphates. Accordingly, growth on Erythritol as the sole C source should require aldolase and fructose-1,6-bisphosphatase to produce essential hexose-6-monophosphate. However, we observed that a mutant devoid of fructose-1,6-bisphosphatases grew normally on Erythritol and that EryC, which was assumed to be a dehydrogenase, actually belongs to the xylose isomerase superfamily. Moreover, we found that TpiA2 and RpiB, distant homologs of triose phosphate isomerase and ribose 5-phosphate isomerase B, were necessary, as previously shown for Rhizobium. By using purified recombinant enzymes, we demonstrated that L-3-tetrulose-4-phosphate was converted to D-erythrose 4-phosphate through three previously unknown isomerization reactions catalyzed by EryC (tetrulose-4-phosphate racemase), TpiA2 (D-3-tetrulose-4-phosphate isomerase; renamed EryH), and RpiB (D-erythrose-4-phosphate isomerase; renamed EryI), a pathway fully consistent with the isotopomer distribution of the erythrose-4-phosphate-derived amino acids phenylalanine and tyrosine obtained from bacteria grown on (13)C-labeled Erythritol. D-erythrose-4-phosphate is then converted by enzymes of the pentose phosphate pathway to glyceraldehyde 3-phosphate and fructose 6-phosphate, thus bypassing fructose-1,6-bisphosphatase.
CONCLUSIONS:
This is the first description to our knowledge of a route feeding carbohydrate metabolism exclusively via D-erythrose 4-phosphate, a pathway that may provide clues to the preferential metabolism of Erythritol by Brucella and its role in pathogenicity. |





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)