| In vivo: |
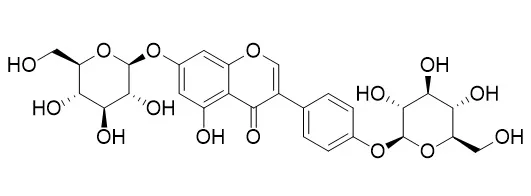
| Plos One, 2014, 9(6):e98559. | | Isolation of Antiosteoporotic Compounds from Seeds of Sophora japonica.[Pubmed: 24892557] | Chemical investigation of Sophora japonica seeds resulted in the isolation of seven metabolites identified as: genistin (1), sophoricoside (2), sophorabioside (3), sophoraflavonoloside (4), Genistein 7,4'-di-O-beta-D-glucopyranoside (5), kaempferol 3-O-α-L-rhamnopyranosyl(1 → 6)β-D-glucopyranosyl(1 → 2)β-D-glucopyranoside (6) and rutin (7).
METHODS AND RESULTS:
Compounds 1, 2 and 5 showed significant estrogenic proliferative effect in MCF-7 cell in sub-cytotoxic concentration range. Compounds 1 and 2 showed minimal cell membrane damaging effect using LDH leakage assay. Accordingly, compound 2 (sophoricoside, (SPH)) was selected for further in-vivo studies as a potential anti-osteoporosis agent. The anti-osteoporotic effect of SPH was assessed in ovarectomized (OVX) rats after oral administration (15 mg/kg and 30 mg/kg) for 45 days compared to estradiol (10 µg/kg) as a positive control. Only in a dose of 30 mg/kg, SPH regained the original mechanical bone hardness compared to normal non-osteoporotic group. However, SPH (15 mg/kg) significantly increased the level of alkaline phosphatase (ALP) to normal level. Treatment with SPH (30 mg/kg) increased the level of ALP to be higher than normal group. SPH (15 mg/kg) did not significantly increase the serum level of osteocalcin (OC) compared to OVX group. On the other hand, treatment with SPH (30 mg/kg) significantly increased the level of OC to 78% higher than normal non-ovarectomized animals group. In addition, SPH (15 mg/kg) decreased the bone resorption marker, acid phosphatase (ACP) to normal level and SPH (30 mg/kg) further diminished the level of serum ACP.
CONCLUSIONS:
Histopathologically, sophoricoside ameliorated the ovarectomy induced osteoporosis in a dose dependent manner. The drug showed thicker bony trabeculae, more osteoid, and more osteoblastic rimming compared to OVX group. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)