| Biosci Biotechnol Biochem. 2008 Sep;72(9):2342-52. |
| Syntheses of all stereoisomers of goniodiol from yeast-reduction products and their antimicrobiological activity.[Pubmed: 18776681] |
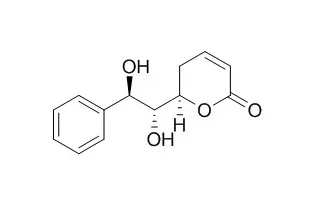
All stereoisomers of Goniodiol were synthesized from yeast-reduction products. The C-6 chiral centers were converted from the chiral centers of the yeast-reduction products.
METHODS AND RESULTS:
Stereoselective conversion of the alkene, which had been prepared from the yeast-reduction product, to glycol constructed the C-7 and C-8 stereochemistry. (+)-Goniodiol and 7-epi-(+)-Goniodiol showed the highest antibacterial activity (MIC, 3.1 mM) against Yersinia intermedia. |
| Org Biomol Chem. 2006 May 7;4(9):1698-706. |
| A highly enantioselective total synthesis of (+)-goniodiol.[Pubmed: 16633562] |
METHODS AND RESULTS:
A high-yielding enantioselective total synthesis of the bioactive styryllactone (+)-Goniodiol has been realised, starting from readily available (S)-glycidol. A key step is an oxygen-to-carbon rearrangement of a silyl enol ether linked via an anomeric centre, facilitating the rapid and diastereoselective construction of this functionalised system. |
| J Org Chem. 2002 Oct 18;67(21):7547-50. |
| Stereoselective syntheses of (+)-goniodiol, (-)-8-epigoniodiol, and (+)-9-deoxygoniopypyrone via alkoxyallylboration and ring-closing metathesis.[Pubmed: 12375995] |
METHODS AND RESULTS:
A convenient synthesis of (+)-Goniodiol, (-)-8-epiGoniodiol, and (+)-9-deoxygoniopypyrone has been developed via asymmetric alkoxyallylboration and ring-closing metathesis pathways. |





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)