| In vitro: |
| Fitoterapia. 2012 Jan;83(1):130-6. | | Iridoid, phenylethanoid and flavonoid glycosides from Sideritis trojana.[Pubmed: 22024633] |
METHODS AND RESULTS:
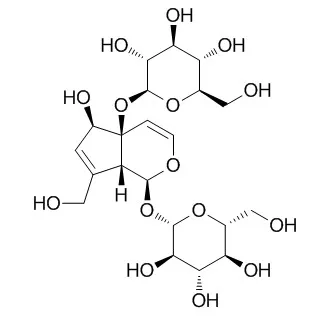
From the MeOH extract of Sideritis trojana, a new iridoid glycoside, 10-O-(E)-feruloylMelittoside (1) was obtained in addition to four known iridoid glycosides [Melittoside (2), 10-O-(E)-p-coumaroylMelittoside (3), stachysosides E (4) and G (5)]. Moreover, five phenylethanoid glycosides [verbascoside (6), isoacteoside (7), lamalboside (8), leonoside A (9), isolavandulifolioside (10), three flavone glycosides (isoscutellarein 7-O-[6'''-O-acetyl-β-allopyranosyl-(1→2)]-β-glucopyranoside (11), 4'-O-methyisoscutellarein 7-O-[6'''-O-acetyl-β-allopyranosyl-(1→2)]-β-glucopyranoside (12), 3'-hydroxy-4'-O-methyisoscutellarein 7-O-[6'''-O-acetyl-β-allopyranosyl-(1→2)]-β-glucopyranoside (13) and a benzylalcohol derivative (di-O-methylcrenatin) were obtained and identified. The structures were elucidated on the basis of NMR and HRMS data.
CONCLUSIONS:
All compounds were tested for their antioxidant activity by in vitro TEAC assay and some of them exhibited moderate activity (0.97-1.44 mM) when compared with the reference compound (quercetin 1.86 mM). Glycosides 6-13, the most active compounds in the TEAC assay, were also tested by flow cytometry to evaluate their ability to affect the levels of reactive oxygen species (ROS) in human prostate cancer cells (PC3). |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)