| In vitro: |
| Nat Prod Res. 2014;28(20):1669-73. | | A new sesquiterpenoid from the rhizomes of Homalomena occulta.[Pubmed: 25104218] |
METHODS AND RESULTS:
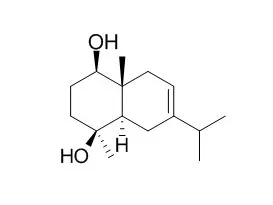
Chemical constituents of EtOAc extract from the rhizomes of traditional Chinese medicine Qian-nian-jian (Homalomena occulta) have been studied, a new sesquiterpenoid, named euadesma-4-ene-1β,15-diol (1), and four related known compounds, polydactin B (2), Oplodiol (3), 1β,4β,7α-trihydroxyeudesmane (4), and (-)1β,4β,6α-trihydroxy-eudesmane (5), were isolated. Their structures were elucidated using spectroscopic methods including 1D and 2D NMR techniques and mass spectrometry. All the isolates were tested against the human lung adenocarcinoma A549 using MTT assay method.
CONCLUSIONS:
Oplodiol (3) and (-)1β,4β,6α-trihydroxy-eudesmane (5) were found to show moderate cytotoxic effects on A549 with IC50 values at 25.5 and 15.0 μg/mL, respectively. | | J Asian Nat Prod Res. 2015 May 12:1-9. | | Sesquiterpenes from the roots of Illicium oligandrum.[Pubmed: 25966312] |
METHODS AND RESULTS:
Two new sesquiterpenes, oligandrin (1) and oligandric acid (2), together with three analogues, tashironin A (3), tashironin (4), and Oplodiol (5), were isolated from the roots of Illicium oligandrum. The structures of new compounds were determined based on 1D and 2D NMR experiments and X-ray diffraction. Compound 1 represents a presumed biosynthetic precursor of seco-prezizaane sesquiterpenes which consists of a novel 6/6/5 tricarbocyclic skeleton. Compound 2 is the first example of chamipinene-type sesquiterpene possessing a 6/4/6 tricyclic system from the genus Illicium. Compounds 1-5 were evaluated in vitro for their activity against coxsackie virus B3 (CVB3), influenza virus A/Hanfang/359/95 (H3N2), and influenza virus A/FM/1/47 (H1N1).
CONCLUSIONS:
Compound 1 showed selective antiviral activity against CVB3 with IC50 value of 11.11 μM. | | Phytochemistry, 1999, 52(6):1095-9. | | Anti-plasmodial sesquiterpenoids from the African Reneilmia cincinnata.[Pubmed: 10643672] |
METHODS AND RESULTS:
A new isodaucane sesquiterpenoid, 6,7,10-trihydoxyisodaucane, was isolated from the fruits of Reneilmia cincinnata, together with the known sesquiterpenoids Oplodiol, oplopanone, 5E,10(14)-germacradien-1 beta, 4 beta-diol, 1(10)E,5E-germacradien-4 alpha-ol and eudesman-1,4,7-triol. A large amount of 5-hydroxy-3,7,4'-trimethoxyflavone was also isolated. Their structures were established by NMR techniques using 1D and 2D experiments.
CONCLUSIONS:
Three of the known sesquirernenoids exhibited noteworthy anti-plasmodial activity against Plasmodium falciparum strains. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)