| Structure Identification: |
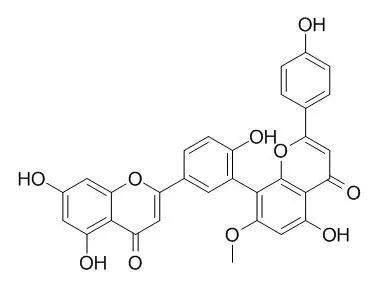
| J Nat Prod. 2003 Jul;66(7):1002-5. | | Chemical constituents from Amentotaxus yunnanensis and Torreyayunnanensis.[Pubmed: 12880325 ] | In a chemical study of taxonomically related Taxaceae plants of Yunnan Province, China, seven compounds, including a new amentoflavone biflavonoid, 2,3-dihydro-7,7' '-dimethoxyamentoflavone (1), were isolated from Amentotaxus yunnanensis, and 12 isolates were obtained from Torreya yunnanensis. From the latter plant, a new abietane diterpene, torreyayunnin (7), is reported for the first time.
METHODS AND RESULTS:
The known isolates from A. yunnanensis have been identified as sequoiaflavone (3), Sotetsuflavone (4), 7,7' '-dimethoxyamentoflavone (5), lutein, beta-sitosterol, and sequoyitol. Amentoflavone (2), Sotetsuflavone (4), sciadopitysin (6), 12-hydroxydehydroabietinol, meridinol, balanophonin, (+)-pinoresinol monomethyl ether, (+)-pinoresinol monomethyl ether glucoside, erythro-1-(4-hydroxy-3-methoxyphenyl)-2-[4-[2-formyl-(E)-vinyl]-2- methoxyphenoxy]propane-1,3-diol, threo-1-(4-hydroxy-3-methoxyphenyl)-2- [4-[2-formyl-(E)-vinyl]-2-methoxyphenoxy] propane-1,3-diol, and (E)-2-butenedioic acid were identified as known isolates from T. yunnanensis.
CONCLUSIONS:
The presence of the amentoflavone biflavonoids (1, 3-5) in A. yunnanensis supports its placement in the Taxaceae. The occurrence of the biflavonoid Sotetsuflavone (4) in both A. yunnanensis and T. yunnanensis suggests that these two genera are closely related. The identification and structural elucidation of these isolates were based on spectral data analysis including 1D and 2D NMR. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)