Natural Products
Eupatilin
| Catalog No. | CFN90190 | 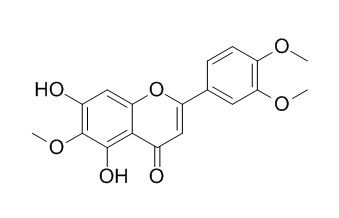 |
| CAS No. | 22368-21-4 | |
| Molecular Weight: | 344.31 | |
| Molecular Formula | C18H16O7 | |
| DBs | [PubChem]:274955023 [ChEMBL]:4932 [PCIDB]:3845 |
Standard InChI:
InChI=1S/C18H16O7/c1-22-12-5-4-9(6-14(12)23-2)13-7-10(19)16-15(25-13)8-11(20)18(24-3)17(16)21/h4-8,20-21H,1-3H3
Biological Activity
Eupatilinthe has anti-proliferative effect in MCF10A- ras cells, is associated with its blockade of cell cycle progression which appears to be attributable in part to inhibition of ERK1/2 activation.[1]
Eupatilin attenuates bile acid-induced hepatocyte apoptosis by suppressing bile acid-induced kinase activation, it might be therapeutically efficacious in a variety of human liver diseases associated with cholestasis.[2]
Eupatilin and jaceosidin have effects on cytochrome p450 enzyme activities in human liver microsomes, have potential pharmacokinetic drug interactions in vivo due to inhibition of CYP1A2 and CYP2C9.[3]
Eupatilin is a potent anti-atherogenic agent that inhibits PDGF-BB-induced proliferation and migration in HASMCs as well as aortic sprouting, which is likely mediated through the attenuation of PI3K, MKK3/6, and MKK4 activation.[4]
Eupatilin suppresses oxidative damage and reciprocally enhances extracellular matrix production in articular chondrocytes, making eupatilin a promising therapeutic option for the treatment.[5]
Eupatilin improves the acute hepatic IRI by reducing inflammation and apoptosis, it is a promising therapeutic agent against acute IR-induced hepatic damage.[6]
Eupatilin protects against tumor necrosis factor-α-mediated inflammation inhuman umbilical vein endothelial cells.[7]
Eupatilin induces Sestrin2-dependent autophagy to prevent oxidative stress.[8]
Product
References
[1] Kim D H, Na H K, Oh T Y, et al. Biochem Pharmacol, 2004, 68(6):1081-7.
[2] Su C P, Yoon J H, Kim W, et al. J Gastroenterol, 2006, 41(8):772-8.
[3] Ji H Y, Kim S Y, Kim D K, et al. Molecules, 2010, 15(9):6466-75.
[4] Son J E, Lee E, Seo S G, et al. Planta Med, 2013, 79(12):1009-16.
[5] Jeong J H, Moon S J, Jhun J Y, et al. Plos One, 2015, 10(6):e0130882.
[6] Lee H M, Jang H J, Kim S S, et al. Transpl P, 2016, 48(4):1226-33.
[7] Yu K, Li X M, Xu X L, et al. Int J Clin Exp Med, 2016, 8(12):22191-7.
[8] Jegal K H, Ko H L, Sang M P, et al. Apoptosis, 2016, 21(5):1-15.
[9] Zhou Q, Sun L L, Jiang B, et al. China Pharm, 2013, 24(47):4464-6.
Product Use Citation





