| In vivo: |
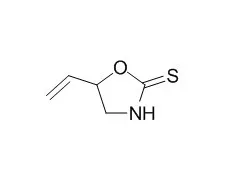
| Chem Senses. 2010 Oct;35(8):685-92. | | Genetics and bitter taste responses to goitrin, a plant toxin found in vegetables.[Pubmed: 20551074] | The perceived bitterness of cruciferous vegetables such as broccoli varies from person to person, but the functional underpinnings of this variation are not known. Some evidence suggests that it arises, in part, from variation in ability to perceive Goitrin (5-vinyloxazolidine-2-thione), a potent antithyroid compound found naturally in crucifers.
METHODS AND RESULTS:
Individuals vary in ability to perceive synthetic compounds similar to Goitrin, such as 6-propyl-2-thiouracil (PROP) and phenylthiocarbamide (PTC), as the result of mutations in the TAS2R38 gene, which encodes a bitter taste receptor. This suggests that taste responses to Goitrin itself may be mediated by TAS2R38. To test this hypothesis, we examined the relationships between genetic variation in TAS2R38, functional variation in the encoded receptor, and threshold taste responses to Goitrin, PROP, and PTC in 50 subjects. We found that threshold responses to Goitrin were associated with responses to both PROP (P = 8.9 x 10(-4); r(s) = 0.46) and PTC (P = 7.5 x 10(-4); r(s) = 0.46). However, functional assays revealed that Goitrin elicits a weaker response from the sensitive (PAV) allele of TAS2R38 (EC(50) = 65.0 μM) than do either PROP (EC(50) = 2.1 μM) or PTC (EC(50) = 1.1 μM) and no response at all from the insensitive (AVI) allele. Furthermore, Goitrin responses were significantly associated with mutations in TAS2R38 (P = 9.3 × 10(-3)), but the same mutations accounted for a smaller proportion of variance in Goitrin response (r(2) = 0.16) than for PROP (r(2) = 0.50) and PTC (r(2) = 0.57).
CONCLUSIONS:
These findings suggest that mutations in TAS2R38 play a role in shaping Goitrin perception, but the majority of variance must be explained by other factors. | | J Nat Prod. 1988 Sep-Oct;51(5):862-5. | | Inhibition of dopamine beta-hydroxylase by goitrin, a natural antithyroid compound.[Pubmed: 2462616] |
METHODS AND RESULTS:
RS-Goitrin can be conveniently prepared by a simplification of the Ettlinger procedure. Goitrin is a moderate inhibitor of purified bovine adrenal dopamine beta-hydroxylase. The administration of Goitrin leads to a depression of brain norepinephrine and to an elevation of heart and adrenal dopamine. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)