| Description: |
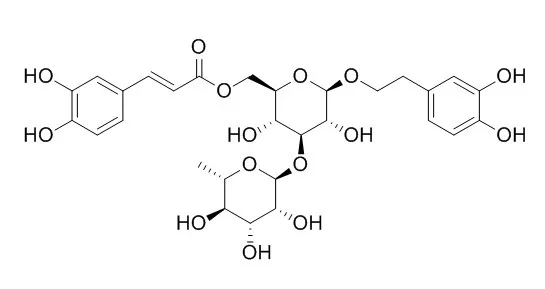
Isoacteoside exhibits significant inhibition of advanced glycation end product formation with IC50 values of 4.6-25.7 μM; it has neuroprotective, antioxidant, and anti-inflammatory effects. Isoacteoside can stimulate the increase of α7 and α3 proteins in the cultured cells, attenuate the decreased expression of α3 and α7 nAChR subunit proteins and cell viability on SH-SY5Y cells induced by Aβ; it can regulate caspase-1, mitogen-activated protein kinases (c-Jun N-terminal kinase, p38, extracellular signal-regulated protein kinase) and nuclear factor-kappa B pathways. |
| In vitro: |
| Immunopharmacol Immunotoxicol. 2015 May 15:1-7. | | Anti-inflammatory effects of isoacteoside from Abeliophyllum distichum.[Pubmed: 25975581] | Isoacteoside, a dihydroxypheynylethyl glycoside, is a major bioactive component of Abeliophyllum distichum (White Forsythia) which is a deciduous shrub native to the south and central areas of Korea.
The present study is designed to evaluate the anti-inflammatory activities and underlying mechanisms of Isoacteoside in human mast cell line, HMC-1 cells.
METHODS AND RESULTS:
We isolated Isoacteoside from A. distichum. The anti-inflammatory effect of Isoacteoside was investigated in HMC-1 cells by studying the following markers: phorbol 12-myristate 13-acetate and calcium ionophore A23187 (PMACI)-induced interleukin (IL)-1β, IL-6, IL-8, and tumor necrosis factor alpha (TNF-α) secretion and mRNA expression by ELISA and RT-PCR, respectively. In addition, mechanism related to anti-inflammatory was investigated by Western blotting. Isoacteoside significantly suppressed the production and mRNA expression of proinflammatory cytokines including IL-1β, IL-6, IL-8 and TNF-α in PMACI-stimulated HMC-1 cells without cytotoxicity.
It was found that anti-inflammatory effects of Isoacteoside are mediated by action on caspase-1, mitogen-activated protein kinases (c-Jun N-terminal kinase, p38, extracellular signal-regulated protein kinase) and nuclear factor-kappa B pathways.
CONCLUSIONS:
Taken together, the present findings provide new insights that Isoacteoside may be a promising anti-inflammatory agent for inflammatory disorders. | | Bot Stud. 2013 Dec;54(1):6. | | Inhibitory activities of acteoside, isoacteoside, and its structural constituents against protein glycation in vitro.[Pubmed: 28510849 ] | Advanced glycation end products (AGE) are substances that can induce insulin resistance in adipocyte, hepatocyte and muscle cells. This resistance correlates highly with cardiovascular disease and diabetic complications. Acteoside (A), a phenylethanoid glycoside, is an active compound in several plants and traditional herbal medicines. Acteoside, its structural isomer, Isoacteoside (I), and their constituents, caffeic acid (C) and 3,4-dihydroxyphenylethanol (D), were used in the study to investigate the inhibitory activity against AGE formations in vitro.
METHODS AND RESULTS:
AGE formations were detected by anti-(Nϵ-(carboxymethyl)lysine (anti-CML), using bovine serum albumin (BSA)/glucose (glc) and BSA/galactose (gal) as models, or by anti-argpyrimidine (anti-AP), using BSA/methylglyoxal (MGO) as models. It was found that A, I, C, or D, each at 5 mM, could attenuate the CML formations detected by ELISA in the BSA/gal model of a 3-day or 5-day reaction, and showed significant differences (P < 0.01 or P < 0.001) compared to the control. However, these compounds showed a minor effect after a 7-day incubation. It was also found that C or D could lower the CML formations in the BSA/glc model and showed significant differences (P < 0.05 or P < 0.01) compared to the control after a 3-day, 5-day and 7-day reaction. It was found that A, I, C, or D, each at 0.5 mM or 5 mM, could attenuate the AP formations in the BSA/MGO model of a 3-day reaction and showed significant differences (P < 0.001) compared to the control.
CONCLUSIONS:
The results suggest the potential anti-glycation activities of A and I in vitro may apply to cell models at higher glucose concentrations or to diabetic animal models, and need further investigation. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)