| Cell Research: |
| Int J Biol Sci. 2016 Jan 1;12(1):72-86. | | Isovitexin Exerts Anti-Inflammatory and Anti-Oxidant Activities on Lipopolysaccharide-Induced Acute Lung Injury by Inhibiting MAPK and NF-κB and Activating HO-1/Nrf2 Pathways.[Pubmed: 26722219] | Oxidative damage and inflammation are closely associated with the pathogenesis of acute lung injury (ALI). Thus, we explored the protective effect of Isovitexin (IV), a glycosylflavonoid, in the context of ALI.
METHODS AND RESULTS:
To accomplish this, we created in vitro and in vivo models by respectively exposing macrophages to lipopolysaccharide (LPS) and using LPS to induce ALI in mice. In vitro, our results showed that IV treatment reduced LPS-induced pro-inflammatory cytokine secretion, iNOS and COX-2 expression and decreased the generation of ROS. Consistent findings were obtained in vivo. Additionally, IV inhibited H2O2-induced cytotoxicity and apoptosis. However, these effects were partially reversed following the use of an HO-1 inhibitor in vitro. Further studies revealed that IV significantly inhibited MAPK phosphorylation, reduced NF-κB nuclear translocation, and upregulated nuclear factor erythroid 2-related factor 2 (Nrf2) and heme oxygenase 1 (HO-1) expression in RAW 264.7 cells. In vivo, pretreatment with IV attenuated histopathological changes, infiltration of polymorphonuclear granulocytes and endothelial activation, decreased the expression of ICAM-1 and VCAM-1, reduced the levels of MPO and MDA, and increased the content of GSH and SOD in ALI. Furthermore, IV treatment effectively increased Nrf2 and HO-1 expression in lung tissues.
CONCLUSIONS:
Therefore, IV may offer a protective role against LPS-induced ALI by inhibiting MAPK and NF-κB and activating HO-1/Nrf2 pathways. | | Planta Med. 2002 Apr;68(4):365-7. | | Prevention of cellular ROS damage by isovitexin and related flavonoids.[Pubmed: 11988866 ] | The antioxidant properties of Isovitexin and related flavonoids were studied.
METHODS AND RESULTS:
Isovitexin inhibited xanthine oxidase with an IC50 value of = 15.2 microM. The flavonoid analogues, apigenin, kaempferol, quercetin, myricetin, and genistein also inhibited xanthine oxidase with IC50 values of 0.58, 2.18, 1.09, 9.90, and 4.83 microM, respectively. Isovitexin protected DNA from the Fenton reaction-induced breakage in a dose-dependent manner with an IC50 value of 9.52 microM. Isovitexin also protected HL-60 cells from the ROS damage induced by the xanthine/xanthine oxidase reaction. Isovitexin exhibited the lowest cytotoxicity toward HL-60 cells (LD50 >400 microM) compared to the other flavonoids examined. In addition, excess hydrogen peroxide induced by cadmium in A2780 ovarian cells was significantly suppressed by Isovitexin.
CONCLUSIONS:
These results suggest that Isovitexin in rice may protect cells from oxidative stress. |
|
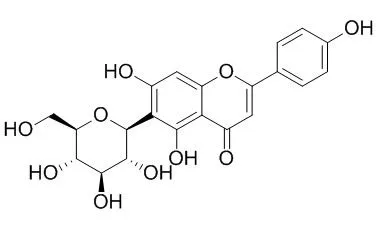
| Structure Identification: |
| J Agric Food Chem. 2012 Oct 31;60(43):10844-9. | | Enzymatic acylation of isoorientin and isovitexin from bamboo-leaf extracts with fatty acids and antiradical activity of the acylated derivatives.[Pubmed: 23057589] |
METHODS AND RESULTS:
This study enzymatically acrylates two flavonoids from bamboo-leaf extracts, isoorientin and Isovitexin, with different fatty acids as acyl donors using Candida antarctica lipase B (CALB).
The conversion yield ranged from 35 to 80% for fatty acids with different chain lengths. Higher isoorientin and Isovitexin conversion yields (>75%) were obtained using lauric acid in tert-amyl-alcohol as the reaction medium. (1)H and (13)C nuclear magnetic resonance spectroscopy analysis showed that, in the presence of CALB, acylation occurred at the isoorientin and Isovitexin primary hydroxyl group of glucose moiety and only monoesters were detected.
CONCLUSIONS:
Introducing an acyl group into isoorientin and Isovitexin significantly improved their lipophilicity but reduced their antiradical activity. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)