| Description: |
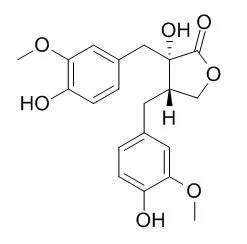
Nortrachelogenin is a novel agent for prostate cancer therapy with ability to inhibit Akt membrane localization and activity as well as the activation of growth factor receptors, thereby efficiently synergizing with tumor necrosis factor-related apoptosis-inducing ligand (TRAIL), exposure.Nortrachelogenin has anti-inflammatory properties,it shows moderate inhibiting activities on NF- κB signaling pathway induced by TNF-α , with the IC50 value of 49.4 uM. Nortrachelogenin also shows anti-plasmodium activity of 14.50 dg/ml. (+ )-Nortrachelogenin shows effects on the central nervous system producing depression in rabbits, it is moderately active against HIV-1 in vitro. (-)-Nortrachelogenin exerts its antibacterial effect by disorganizing and perturbing the cytoplasmic membrane, it also can induce membrane disruption and caspase-dependent apoptosis. |
| In vitro: |
| Int. J.Appl. Res.Nat.Prod.,2011,4(3):755-6. | | Anti-plasmodial activity of Nortrachelogenin from the root bark of Carissa edulis (vahl)[Reference: WebLink] |
METHODS AND RESULTS:
The decoction of the root bark of Carrisa edulis is used traditionally for treatment of malaria and other ailments. Plasmodium falcipurum in vitro drug sensitive study was conducted in order to evaluate the correlation between the ethno medicinal use and bioactivity of fractions and total extract of the plant. Methanolic extract of the root bark of carissa edulis showed anti-plasmodial activity against the chloroquin-senitive (D6) strains of plasmodium falciparum parasite with IC50 value of 1.95 Dg/ml.
CONCLUSIONS:
From this extract, a lignan compound Nortrachelogenin was isolated and showed anti-plasmodium activity of 14.50 Dg/ml. The structure was determined on the basis of spectroscopic evidence. | | FEMS Yeast Res. 2016 May;16(3). | | (-)-Nortrachelogenin from Partrinia scabiosaefolia elicits an apoptotic response in Candida albicans.[Pubmed: 26880798 ] | This study analyzes the antifungal properties of (-)-Nortrachelogenin and elucidates its mode of action against pathogenic fungi.
METHODS AND RESULTS:
We performed susceptibility tests against several pathogenic fungi and verified the absence of hemolysis against human erythrocytes. Its antifungal activity increased reactive oxygen species (ROS) in response to intracellular stress and increased concentrations of both intracellular and extracellular trehalose without causing hemolysis. In addition, a cell wall regeneration study indicated its action on the cytoplasmic membrane. A cell surface study using 3,3(')-dipropylthiacarbocyanine iodide [DiSC3(5)] and 1,6-diphenyl-1,3,5-hexatriene (DPH) demonstrated dissipation of the cytoplasmic membrane at high concentrations. Our study revealed a disturbance in the membrane at higher concentrations and externalization of phosphatidylserine in a dose-dependent manner, affecting other intracellular responses. Furthermore, we investigated the late stage of apoptosis using TUNEL and 4('),6-diamidino-2-phenylindole (DAPI) assays.
CONCLUSIONS:
(-)-Nortrachelogenin-treated cells underwent apoptosis which was triggered by mitochondrial dysfunction via depolarization of the mitochondrial membrane, release of cytochrome c and calcium ion signaling, resulting in the activation of metacaspases. Different concentrations of (-)-Nortrachelogenin induced membrane disruption and caspase-dependent apoptosis. | | Curr Microbiol. 2016 Jan;72(1):48-54. | | Antibacterial Mechanism of (-)-Nortrachelogenin in Escherichia coli O157.[Pubmed: 26420306 ] | (-)-Nortrachelogenin is a lignan belonging to group of polyphenolic compounds. Its biological properties in mammalian cells are well-studied; however, its biological effects in microorganisms remain poorly understood. Its efficacy against pathogenic bacteria, including antibiotic-resistant strains, was investigated and it was found that bacteria are highly susceptible to the antibacterial effects of this compound.
METHODS AND RESULTS:
To investigate the antibacterial mode of action(s) against Escherichia coli O157, its effect on the penetration of SYTOX green into bacterial cells was assayed. The penetration of SYTOX Green into a bacterial cell is a measure of permeability of the plasma membrane. An increase in fluorescence intensity using bis-(1,3-dibutylbarbituric acid) trimethine oxonol [DiBAC4(3)] and 3,3'-dipropylthiacarbocyanine iodide [DiSC3(5)] was also observed, indicating membrane depolarization. Potassium ion efflux from the cytosol into the extracellular matrix showed that cellular damage due to (-)-Nortrachelogenin treatment resulted in the loss of intracellular components. While cells were damaged by (-)-Nortrachelogenin, large unilamellar vesicles containing fluorescein isothiocyanate-dextran were perturbed to migrate molecules between 3.3 and 4.8 nm. The release of calcein from giant unilamellar vesicles, occurring as a result of disruption in artificial membranes, was visualized.
CONCLUSIONS:
Taken together, our results indicate that (-)-Nortrachelogenin exerts its antibacterial effect by disorganizing and perturbing the cytoplasmic membrane, demonstrating the potential of this compound as a candidate for antibiotic drug development. | | Planta Med. 2000 Aug;66(6):564-7. | | Antifungal, antimitotic and anti-HIV-1 agents from the roots of Wikstroemia indica.[Pubmed: 10985087 ] |
METHODS AND RESULTS:
With guidance of Pyricularia oryzae bioassay, daphnoretin (1), (+)-Nortrachelogenin (2), genkwanol A (3), wikstrol A (4), wikstrol B (5) and daphnodorin B (6) were isolated from the roots of Wikstroemia indica. Compounds 1-6 induced morphological deformation of P. oryzae mycelia with MMDC values of 68.4 +/- 1.3, 31.3 +/- 1.8, 45.8 +/- 0.5, 70.1 +/- 2.4, 52.3 +/- 0.9 and 73.7 +/- 1.6 microM, respectively. Compounds 3-6 showed moderate activity against microtubule polymerization with IC50 values of 112 +/- 4, 131 +/- 3, 184 +/- 6 and 142 +/- 2 microM in vitro, respectively. Compounds 2, 3, 5 and 6 were moderately active against HIV-1 in vitro.
CONCLUSIONS:
The findings of bioactivity of 1-6 support the antifungus, antimitosis and anti-HIV-1 uses for W. indica roots. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)