| In vitro: |
| Food Chem., 2012, 133(3):998-1000. | | Cyclooxygenase inhibitory activity of ginsenosides from heat-processed ginseng.[Reference: WebLink] | Ginsenosides, from heat-processed ginseng, and sapogenins were evaluated for their inhibitory effects on the enzyme catalytic activities of cyclooxygenases-1 and -2 (COX-1 and -2).
METHODS AND RESULTS:
The ginsenosides, 20(S)-Rg3, Rg5, and Rk1, inhibited COX-2 activity, but did not affect the enzyme activity of COX-1. Protopanaxatriol (PPT) moderately inhibited both COX-1 and -2. The ginsenosides, 20(R)-Rg3, Re, and protopanaxadiol (PPD) showed a minimal effect on COX-1 and -2 activities.
CONCLUSIONS:
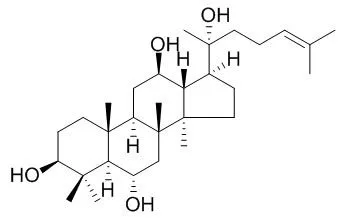
Taken together, ginsenosides Rg3 (20S-form), Rg5 and Rk1 showed selective inhibitory activity on COX-2 by behaving as an inhibitor of the enzyme–substrate reaction. | | Chemical Research in Chinese Universities, 2016, 32(1): 1-6. | | Semisynthesis and cytotoxicity evaluation of a series of ocotillol type saponins and aglycones from 20(S)-ginsenoside Rg2, Rh1, protopanaxatriol and their 20(R)-epimers[Reference: WebLink] | With the oxidation treatment, eighteen compounds were separated from 20(S)-ginsenoside Rg2, Rh1, protopanaxatriol(PPT) and their 20(R)-epimers in total and cytotoxicity of most of them was evaluated against three human cancer cell lines HeLa, A549 and MCF-7 by 3-(4,5-dimetylthiazol-z-yl)-2,5-diphenyltetrazolium bromide(MTT) assay.
METHODS AND RESULTS:
Their structures were confirmed by means of nuclear magnetic resonance(NMR) and mass spectrometry and the results were compared with those of previous literature. In this study, we systematically semisynthesized all four ocotillol type saponins, i.e., (20S, 24S), (20S, 24R), (20R, 24S) and (20R, 24R). All the configurations at C20 kept the same with their starting materials. Meanwhile a pair of C24 epimers was generated in terms of ocotillol type saponins. In addition, seven compounds(4―8, 13 and 14) were reported firstly.
CONCLUSIONS:
The cytotoxic results distinguished the ocotillol type products(6, 7, 13 and 14) from 20(R)-Protopanaxatriol and 20(R)-ginsenoside Rh1, which possessed better cytotoxicities than their correspondents from 20(S)-epimers against HeLa cells, and the carbonyl group at C3 can improve the cytotoxicity, which helped us to gain deeper insight into Ocotillol type saponins. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)