| Description: |
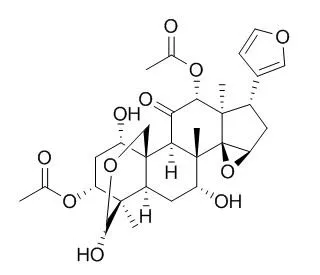
Toosendanin (TSN) was used as a digestive tract-parasiticide and agricultural insecticide in ancient China;TSN is a selective presynaptic blocker, a L-type Ca 2+ channel agonist and an effective antibotulismic agent, by interfering with neurotransmitter release through an initial facilitation followed by a subsequent depression. TSN has effects on the growth, cell cycle arrest, induction of apoptosis and the involved signaling pathway in human promyelocytic leukemia HL-60 cells. |
| Targets: |
Calcium Channel | JNK | Bcl-2/Bax | PARP | Caspase |
| In vitro: |
| Prog Neurobiol. 2007 May;82(1):1-10. | | Biological effects of toosendanin, a triterpenoid extracted from Chinese traditional medicine.[Pubmed: 17363132 ] | Toosendanin (TSN) is a triterpenoid extracted from Melia toosendan Sieb et Zucc, which was used as a digestive tract-parasiticide and agricultural insecticide in ancient China. TSN was demonstrated to be a selective presynaptic blocker and an effective antibotulismic agent.
METHODS AND RESULTS:
By interfering with neurotransmitter release through an initial facilitation followed by a subsequent depression, TSN eventually blocks synaptic transmission at both the neuro-muscular junction and central synapses. Despite sharing some similar actions with botulinum neurotoxin (BoNT), TSN has a marked antibotulismic effect in vivo and in vitro. Studies suggest that the antibotulismic effect of TSN is achieved by preventing BoNT from approaching its enzymatic substrate, the SNARE protein. It is also found that TSN can induce differentiation and apoptosis in several cell lines, and suppress proliferation of various human cancer cells. TSN inhibits various K(+)-channels, selectively facilitates Ca(2+)-influx via L-type Ca(2+) channels and increases intracellular Ca(2+) concentration ([Ca(2+)](i)).
CONCLUSIONS:
The TSN-induced [Ca(2+)](i) increase and overload could be responsible for the TSN-induced biphasic effect on transmitter release, cell differentiation, apoptosis as well as the cytoxicity of TSN. | | J Med Entomol. 2013 Jan;50(1):112-21. | | Effects of the botanical insecticide, toosendanin, on blood digestion and egg production by female Aedes aegypti (Diptera: Culicidae): topical application and ingestion.[Pubmed: 23427659] | Botanical insecticides offer novel chemistries and actions that may provide effective mosquito control.
METHODS AND RESULTS:
Toosendanin (TSN, 95% purity) is one such insecticide used to control crop pests in China, and in this study, it was evaluated for lethal and sublethal effects on larvae and females of the yellowfever mosquito, Aedes aegypti (L.). Toosendanin was very toxic to first instar larvae after a 24 h exposure (LC50 = 60.8 microg/ml) and to adult females up to 96 h after topical treatment (LD50 = 4.3 microg/female) or ingestion in a sugar bait (LC50 = 1.02 microg/microl). Treatment of first instars for 24 h with a range of sublethal doses (6.3-25 microg/ml) delayed development to pupae by 1 to 2 d. Egg production and larval hatching from eggs were dose dependently reduced (>45%) by Toosendanin doses (1.25-10.0 microg) topically applied to females 24 h before or 1 h after a bloodmeal. Ingestion of Toosendanin (0.031-0.25 microg/microl of sugar bait) by females 24 h before a bloodmeal also greatly reduced egg production and larval hatch; no eggs were oviposited by females ingesting the highest dose. Further studies revealed that topical or ingested Toosendanin dose-dependently disrupted yolk deposition in oocytes, blood ingestion and digestion, and ovary ecdysteroid production in blood-fed females.
CONCLUSIONS:
Overall, our results indicate that Toosendanin is an effective insecticide for Ae. aegypti larvae and adults, because of its overt toxicity at high doses and disruption of development and reproduction at sublethal doses. |
|
| In vivo: |
| J. Appl. Entomol., 1995, 119(1-5):367-70. | | Antifeedant and growth inhibitory effects of the limonoid toosendanin and Melia toosendan extracts on the variegated cutworm, Peridromasaucia (Lep., Noctuidae)[Reference: WebLink] | Antifeedant and growth inhibitory effects of Toosendanin, a limonoid allelochemical from the bark of the trees Melia toosendan and M. azedarach, were determined for the variegated cutworm, Peridroma saucia, using different bioassays.
METHODS AND RESULTS:
Toosendanin significantly deterred feeding of 2nd and 4th instar larvae in diet choice and leaf disc choice bioassays, respectively. Refined bark extracts containing 60–75% Toosendanin were less deterrent than the pure compound in both bioassays. However, the 60% extract significantly inhibited growth of neonate larvae in a dose-dependent manner when incorporated into an artificial diet, with an EC50 (concentration reducing growth by 50% relative to controls) of 42.3 ppm at 7 days. Extracts containing 60% and 75% Toosendanin were more inhibitory than equivalent amounts of pure Toosendanin in this chronic growth bioassay, suggesting the presence of other minor constituents which must be significantly more inhibitory, though less deterrent to feeding, than Toosendanin itself.
CONCLUSIONS:
Nutritional analyses of 4th instar larvae following both oral and topical administration of Toosendanin or the 75% extract confirm that both materials possess a combination of antifeedant and growth inhibitory properties. | | Int Immunopharmacol . 2019 Nov;76:105909. | | Toosendanin alleviates dextran sulfate sodium-induced colitis by inhibiting M1 macrophage polarization and regulating NLRP3 inflammasome and Nrf2/HO-1 signaling[Pubmed: 31520988] | | Abstract
Toosendanin (TSN), a triterpenoid extracted from the bark of fruit of Melia toosendan Sieb et Zucc, has been proven to have various biological activities including anti-inflammatory activity. But its effects on experimental colitis remain unreported. Herein, we investigated the role and potential mechanisms of TSN in dextran sulfate sodium (DSS) induced colitis in mice. The results showed that, TSN reduced colitis-associated disease activity index (DAI), shortened colon length, and weakened the pathological damage of the colon tissues in murine colitis models. Further studies disclosed that, TSN inhibited the secretion of proinflammatory cytokines and oxidative stress, and suppressed M1 macrophage polarization and the activation of NLR family pyrin domain containing 3 (NLRP3) inflammasome, but upregulated HO-1/Nrf2 expression in murine colitis. In addition, TSN maintained intestinal barrier by regulating zonula occludens-1 (ZO-1) and occludin expression. In conclusion, our findings demonstrated that, TSN alleviates DSS-induced experimental colitis by inhibiting M1 macrophage polarization and regulating NLRP3 inflammasome and Nrf2/HO-1 signaling, and may provide a novel Chinese patent medicine for the treatment of murine colitis.
Keywords: Colitis; Intestinal barrier; Macrophage; NLRP3 inflammasome; Nrf2/HO-1; Toosendanin. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)