| Kinase Assay: |
| Cancers, 2019,11(3):427. | | The Flavonoid Metabolite 2,4,6-Trihydroxybenzoic Acid Is a CDK Inhibitor and an Anti-Proliferative Agent: A Potential Role in Cancer Prevention.[Reference: WebLink] | Flavonoids have emerged as promising compounds capable of preventing colorectal cancer (CRC) due to their anti-oxidant and anti-inflammatory properties. It is hypothesized that the metabolites of flavonoids are primarily responsible for the observed anti-cancer effects owing to the unstable nature of the parent compounds and their degradation by colonic microflora.
METHODS AND RESULTS:
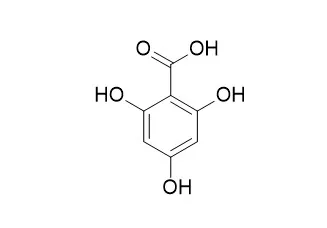
In this study, we investigated the ability of one metabolite, 2,4,6-Trihydroxybenzoic acid (2,4,6-THBA) to inhibit Cyclin Dependent Kinase (CDK) activity and cancer cell proliferation. Using in vitro kinase assays, we demonstrated that 2,4,6-THBA dose-dependently inhibited CDKs 1, 2 and 4 and in silico studies identified key amino acids involved in these interactions. Interestingly, no significant CDK inhibition was observed with the structurally related compounds 3,4,5-trihydroxybenzoic acid (3,4,5-THBA) and phloroglucinol, suggesting that orientation of the functional groups and specific amino acid interactions may play a role in inhibition. We showed that cellular uptake of 2,4,6-THBA required the expression of functional SLC5A8, a monocarboxylic acid transporter. Consistent with this, in cells expressing functional SLC5A8, 2,4,6-THBA induced CDK inhibitory proteins p21Cip1 and p27Kip1 and inhibited cell proliferation.
CONCLUSIONS:
These findings, for the first time, suggest that the flavonoid metabolite 2,4,6-THBA may mediate its effects through a CDK- and SLC5A8-dependent pathway contributing to the prevention of CRC. |
|
| Structure Identification: |
| China Journal of Chinese Materia Medica, 2001, 26(4):260-262. | | Studies on chemical constitutents from Penthorum chinense Pursh.[Reference: WebLink] | To study the chemical constituents of Penthorum chinense.
METHODS AND RESULTS:
Silica gel and macroporous resin were used as adsorbent for isolation and purification. The chemical structures were elucidated by spectral analysis and chemical methods.Three compounds were isolated from the water-extract of P. chinense. The structures were identified as 5-hydroxy-flavanone-7-O-β-D-glucoside (Ⅰ), 2,4,6-Trihydroxybenzoic acid (Ⅱ) and quercetin(Ⅲ), respectively.
CONCLUSIONS:
Compounds Ⅰ and Ⅱ were isolated from this plant for the first time. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)