| Description: |
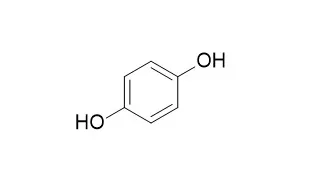
Hydroquinone is a nonvolatile chemical used in the photographic, rubber, chemical, and cosmetic industries. Hydroquinone is one of the most effective inhibitors of melanogenesis in vitro and in vivo, and is widely used for the treatment of melanosis and other hyperpigmentary disorders, the depigmenting activity of it may partly be related to the ability of the compound to act as an alternate substrate of tyrosinase, thereby competing for tyrosine oxidation in active melanocytes. |
| In vitro: |
| Biochimica Et Biophysica Acta General Subjects, 1991,1073(1): 85-90. | | Mechanism of inhibition of melanogenesis by hydroquinone.[Reference: WebLink] | Hydroquinone (HQ) is one of the most effective inhibitors of melanogenesis in vitro and in vivo, and is widely used for the treatment of melanosis and other hyperpigmentary disorders.
METHODS AND RESULTS:
In an attempt to get some insight into the molecular mechanism of the depigmenting action, which is still very poorly understood, we have investigated the effect of HQ on the tyrosinase catalysed conversion of tyrosine to melanin. Incubation of 0.5 mM tyrosine with 0.07 U/ml tyrosinase in phosphate buffer at pH 6.8 in the presence of 0.5 mM HQ led to no detectable melanin formation, due to the preferential oxidation of HQ with respect to tyrosine (HPLC evidence). Kinetic investigations showed that HQ is a poorer substrate of tyrosinase than tyrosine; yet, it may be effectively oxidised in the presence of tyrosine owing to the generation of catalytic amounts of dopa acting as cofactor of tyrosinase. Product analysis of HQ oxidation with tyrosinase in the presence of dopa showed the predominant formation in the early stages of hydroxybenzoquinone (HBQ), arising from enzymic hydroxylation and subsequent oxidation of HQ, along with lower amounts of benzoquinone (BQ).
CONCLUSIONS:
These results suggest that the depigmenting activity of HQ may partly be related to the ability of the compound to act as an alternate substrate of tyrosinase, thereby competing for tyrosine oxidation in active melanocytes. |
|
| In vivo: |
| J Toxicol Environ Health, 1996, 47(1):31-46. | | Human exposure to naturally occurring hydroquinone.[Pubmed: 8568910] | Hydroquinone (HQ) is a nonvolatile chemical used in the photographic, rubber, chemical, and cosmetic industries. HQ is also known to occur in nature as the beta-D-glucopyranoside conjugate (arbutin), and free HQ is a known component of cigarette smoke. Low concentrations of HQ have been detected in the urine and plasma of humans with no occupational or other known exposure to HQ.
METHODS AND RESULTS:
The studies reported here investigate dietary and other potential sources of HQ and their contribution to HQ concentrations in the plasma and urine of human volunteers. Analysis of possible food sources of HQ by GC indicated significant amounts of arbutin in wheat products (1-10 ppm), pears (4-15 ppm), and coffee and tea (0.1 ppm). Free HQ was found in coffee (0.2 ppm), red wine (0.5 ppm), wheat cereals (0.2-0.4 ppm), and broccoli (0.1 ppm). After consuming a meal including arbutin- and HQ-containing foods, volunteers showed significant increases in plasma and urinary levels of HQ and its conjugated metabolites (total HQ). Mean plasma concentrations of total HQ peaked at 5 times background levels at 2 h after the completion of the meal, and mean urinary excretion rates of total HQ peaked at 12 times background at 2-3 h after the meal. Immediately after smoking four cigarettes in approximately 30 min, mean plasma concentrations of total HQ were maximally 1.5 times background levels; mean urinary excretion rates of total HQ peaked at 2.5 times background at 1-3 h after smoking.
CONCLUSIONS:
These data indicate that considerable human exposure to HQ can result from plant-derived dietary sources and, to a lesser extent, from cigarette smoke. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)