| In vitro: |
| Parasitol Res. 2014 Sep;113(9):3381-92. Epub 2014 Jul 13. | | cture–function relationships of inhibition of mosquito cytochrome P450 enzymes by flavonoids of Andrographis paniculata.[Pubmed: 25015047] | The cytochrome P450 monooxygenases are known to play a major role in pyrethroid resistance, by means of increased rate of insecticide detoxification as a result of their overexpression. Inhibition of detoxification enzymes may help disrupting insect detoxifying defense system. The Anopheles minimus CYP6AA3 and CYP6P7 have shown pyrethroid degradation activity and been implicated in pyrethroid resistance.
METHODS AND RESULTS:
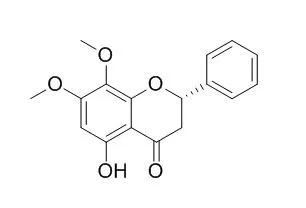
In this study inhibition of the extracts and constituents of Andrographis paniculata Nees. leaves and roots was examined against benzyloxyresorufin O-debenzylation (BROD) of CYP6AA3 and CYP6P7. Four purified flavones (5,7,4′-trihydroxyflavone, 5-hydroxy-7,8-dimethoxyflavone, 5-hydroxy-7,8,2′,3′-tetramethoxyflavone, and 5,4′-dihydroxy-7,8,2′,3′-tetramethoxyflavone), one flavanone (5-Hydroxy-7,8-dimethoxyflavanone) and a diterpenoid (14-deoxy-11,12-didehydroandrographolide) containing inhibitory effects toward both enzymes were isolated from A. paniculata. Structure–function relationships were observed for modes and kinetics of inhibition among flavones, while diterpenoid and flavanone were inferior to flavones. Docking of flavones onto enzyme homology models reinforced relationships on flavone structures and inhibition modes. Cell-based inhibition assays employing 3-(4,5-dimethylthiazol-2-y-l)-2,5-diphenyltetrazolium bromide (MTT) cytotoxicity assays revealed that these flavonoids efficiently increased susceptibility of CYP6AA3- and CYP6P7-expressing Spodoptera frugiperda (Sf9) cells to cypermethrin toxicity, due to inhibition effects on mosquito enzymes.
CONCLUSIONS:
Thus synergistic effects on cypermethrin toxicity of A. paniculata compounds as a result of enzyme inhibition could be useful for mosquito vector control and insecticide resistance management in the future. | | J Agric Food Chem. 2010 Feb 24;58(4):2505-12. | | Anti-inflammatory activity of new compounds from Andrographis paniculata by NF-kappaB transactivation inhibition.[Pubmed: 20085279] | Previous studies showed that the ethyl acetate (EtOAc) fraction of Andrographis paniculata (AP) possessed anti-inflammatory activity.
METHODS AND RESULTS:
This study further isolated these active compounds from bioactivity-guided chromatographic fractionation and identified eight pure compounds. Reporter gene assay indicated that 5-hydroxy-7,8-dimethoxyflavone (1), 5-Hydroxy-7,8-dimethoxyflavanone (2), a mix of beta-sitosterol (3a) and stigmasterol (3b), ergosterol peroxide (4), 14-deoxy-14,15-dehydroandrographolide (5), and a new compound, 19-O-acetyl-14-deoxy-11,12-didehydroandrographolide (6a), significantly inhibited the transcriptional activity of NF-kappaB in LPS/IFN-gamma stimulated RAW 264.7 macrophages (P < 0.05). The two most abundant compounds, 14-deoxy-11,12-didehydroandrographolide (7) and andrographolide (8), had less inhibitory activity but exerted greater inhibitory activity by hydrogenation, oxidation, or acetylation to become four derived compounds, 9, 10, 11, and 12.
CONCLUSIONS:
All of the compounds significantly decreased TNF-alpha, IL-6, macrophage inflammatory protein-2 (MIP-2), and nitric oxide (NO) secretions from LPS/IFN-gamma stimulated RAW 264.7 cells. Compounds 5, 11, and 12 exerted the strongest inhibitory effect on NF-kappaB-dependent transactivation in the RAW 264.7 cell, with IC(50) values of 2, 2.2, and 2.4 microg/mL, respectively, providing encouraging results for bioactive compound development. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)