| In vitro: |
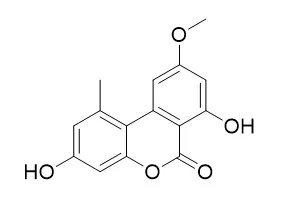
| Journal of Agricultural & Food Chemistry, 1989, 37(5):1341-1343. | | Isolation, identification, and mutagenicity of alternariol monomethyl ether.[Reference: WebLink] | Alternariol monomethyl ether (AME), an Alternaria toxin, was isolated from the media of Alternaria alternata strain 261, a fungus from grains of Linxian County, China, an area noted for a high incidence of esophageal cancer. AME was purified by silica gel chromatography and recrystallized from acetone.
METHODS AND RESULTS:
The chemical structure was identified by systematic spectral analysis. The 60-MHz proton magnetic resonance spectrum exhibited long-distance couplings between 6′-CH3 and 5′,6-hydrogens, and the assignments to two pairs of meta-coupled aromatic hydrogens were revised. Decoupling experiments supported the new assignments. Irradiation at 2.77 ppm peak (6′-CH3) collapsed the 7.28 ppm dull doublet into sharp peaks and changed the 6.77 ppm diquadruplet into a sharp doublet, while peaks at 6.55 and 6.69 ppm remained unchanged. Therefore, the peaks at 6.55, 7.28, 6.69, and 6.77 ppm should be 3′-, 5′-, 4-, and 6-hydrogens, respectively. AME was a strong mutagen in Escherichia coli strain ND-160. Compared to spontaneous revertants, the number of revertants caused by 50 and 100 μg of AME/plate was 6- and 10-fold greater, respectively.
CONCLUSIONS:
Since Scott reported in 1980 that the mutagenicity of AME to Salmonella typhimurium TA98 was weak or marginal, it appears that the mutagenic action of AME exhibits some selectivity toward specific genomic regions or DNA sequences. | | Toxicol Lett. 2012 Oct 2;214(1):46-52. | | Minor contribution of alternariol, alternariol monomethyl ether and tenuazonic acid to the genotoxic properties of extracts from Alternaria alternata infested rice.[Pubmed: 22906495 ] | Alternaria spp. are known to form a spectrum of secondary metabolites with alternariol (AOH), Alternariol monomethyl ether (AME), altenuene (ALT) and tenuazonic acid (TA) as the major mycotoxins with respect to quantity.
METHODS AND RESULTS:
In the present study we investigated the contribution of these compounds for the DNA damaging properties of complex extracts of Alternaria spp. infested rice. Five different Alternaria strains were cultured on rice and analyzed for their production of AOH, AME, ALT and TA. The extracts of two strains with distinctly different toxin profiles were selected for further toxicological analysis. An extract from A. alternata DSM 1102 infested rice, found to contain predominantly TA, exhibited substantial DNA strand breaking properties in cultured human colon carcinoma cells in the comet assay, whereas TA as a single compound did not affect DNA integrity up to 200μM. An extract of A. alternata DSM 12633 infested rice, containing in comparable proportions AOH, AME and TA, exceeded by far the DNA damaging properties of the single compounds. In contrast to AOH, AME and TA, both selected extracts induced an increase of DNA modifications sensitive to the bacterial repair enzyme formamidopyrimidine DNA glycosylase (FPG) in the comet assay, indicative for oxidative DNA damage. Toxicity-guided fractionation of the DSM 12633 extract confirmed that these effects were not caused by AOH, AME or TA.
CONCLUSIONS:
Taken together, the mycotoxins AOH, AME and TA, representing the major mycotoxins with respect to quantity in A. alternata infested food, play only a subordinate role for the genotoxic properties of complex extracts and appear not to be involved in the induction of FPG sensitive sites. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)