| In vitro: |
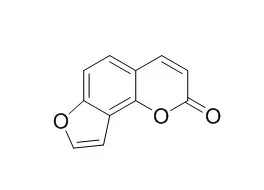
| J Surg Res. 2013 Nov;185(1):300-9. | | Angelicin regulates LPS-induced inflammation via inhibiting MAPK/NF-κB pathways.[Pubmed: 23816246] | Angelicin is a furocoumarin found in Psoralea corylifolia L. fruit. The purpose of this study was to investigate the protective ability of Angelicin against inflammation in lipopolysaccharide (LPS)-stimulated RAW 264.7 cells and LPS-induced in vivo acute lung injury model.
METHODS AND RESULTS:
The concentrations of tumor necrosis factor alpha (TNF-α) and interleukin (IL)-6 in the culture supernatants of RAW 264.7 cells were determined 24 h after LPS administration. ALI was induced by intratracheal instillation of LPS. Six hours after LPS inhalation, bronchoalveolar lavage fluid and lung tissue samples were obtained for enzyme-linked immunosorbent assay, histologic, and Western blotting analyses.
The results showed that pretreatment with Angelicin markedly downregulated TNF-α and IL-6 levels in vitro and in vivo, and significantly decreased the amount of inflammatory cells, lung wet-to-dry weight ratio, and myeloperoxidase activity in LPS-induced ALI mice. Furthermore, Western blotting analysis results demonstrated that Angelicin blocked the phosphorylation of IκBα, NF-κBp65, p38 MAPK, and JNK in LPS-induced ALI.
CONCLUSIONS:
These results suggest that Angelicin was potentially advantageous to prevent inflammatory diseases by inhibiting NF-κB and MAPK pathways. Our data indicated that Angelicin might be a potential new agent for prevention of inflammatory reactions and diseases in the clinic.
| | J. Med. Chem., 2010, 53(4):1519-33. | | Anti-influenza drug discovery: structure-activity relationship and mechanistic insight into novel angelicin derivatives.[Pubmed: 20092255 ] |
METHODS AND RESULTS:
By using a cell-based high throughput screening campaign, a novel Angelicin derivative 6a was identified to inhibit influenza A (H1N1) virus induced cytopathic effect in Madin-Darby canine kidney cell culture in low micromolar range. Detailed structure-activity relationship studies of 6a revealed that the Angelicin scaffold is essential for activity in pharmacophore B, while meta-substituted phenyl/2-thiophene rings are optimal in pharmacophore A and C. The optimized lead 4-methyl-9-phenyl-8-(thiophene-2-carbonyl)-furo[2,3-h]chromen-2-one (8g, IC(50) = 70 nM) showed 64-fold enhanced activity compared to the high throughput screening (HTS) hit 6a. Also, 8g was found effective in case of influenza A (H3N2) and influenza B virus strains similar to approved anti-influenza drug zanamivir (4).
CONCLUSIONS:
Preliminary mechanistic studies suggest that these compounds act as anti-influenza agents by inhibiting ribonucleoprotein (RNP) complex associated activity and have the potential to be developed further, which could form the basis for developing additional defense against influenza pandemics. | | Eur J Haematol. 2003 Sep;71(3):189-95. | | Accumulation of gamma-globin mRNA in human erythroid cells treated with angelicin.[Pubmed: 12930320] |
The aim of the present study was to determine whether Angelicin is able to increase the expression of gamma-globin genes in human erythroid cells. Angelicin is structurally related to psoralens, a well-known chemical class of photosensitizers used for their antiproliferative activity in treatment of different skin diseases (i.e., psoriasis and vitiligo).
METHODS AND RESULTS:
To verify the activity of Angelicin, we employed two experimental cell systems, the human leukemic K562 cell line and the two-phase liquid culture of human erythroid progenitors isolated from normal donors. The results of our investigation suggest that Angelicin, compared with cytosine arabinoside, mithramycin and cisplatin, is a powerful inducer of erythroid differentiation and gamma-globin mRNA accumulation of human leukemia K562 cells. In addition, when normal human erythroid precursors were cultured in the presence of Angelicin, increases of gamma-globin mRNA accumulation and fetal hemoglobin (HbF) production, even higher than those obtained using hydroxyurea, were detected.
CONCLUSIONS:
These results could have practical relevance, as pharmacologically-mediated regulation of the expression of human gamma-globin genes, leading to HbF induction, is considered a potential therapeutic approach in hematological disorders, including beta-thalassemia and sickle cell anemia. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)