| In vivo: |
| BioFactors (Oxford, England)2000, 13(1-4):225-230 | | Anti-tumor and anti-carcinogenic activities of triterpenoid, beta-boswellic acid.[Reference: WebLink] |
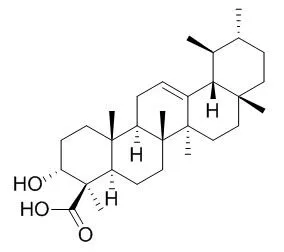
Boswellin (BE), a methanol extract of the gum resin exudate of Boswellia serrata, contains naturally occurring triterpenoids, Beta-boswellic acid and its structural related derivatives, has been used as a traditional medicine for the treatment of inflammatory and arthritic diseases.
METHODS AND RESULTS:
Topical application of BE to the backs of mice markedly inhibited 12-O-tetradecanoylphorbol-13-acetate (TPA)-induced increases in skin inflammation, epidermal proliferation, the number of epidermal cell layers, and tumor promotion in 7,12-dimethylbenz[a]anthracene (DMBA)-initiated mice. Feeding 0.2% of BE in the diet to CF-1 mice for 10-24 weeks reduced the accumulation of parametrial fat pad weight under the abdomen, and inhibited azoxymethane (AOM)-induced formation of aberrant crypt foci (ACF) by 46%. Addition of pure Beta-boswellic acid, 3-O-acetyl-Beta-boswellic acid, 11-keto-Beta-boswellic acid or 3-O-acetyl-11-keto-Beta-boswellic acid to human leukemia HL-60 cell culture inhibited DNA synthesis in HL-60 cells in a dose-dependent manner with IC50 values ranging from 0.6 to 7.1 microM.
CONCLUSIONS:
These results indicate that Beta-boswellic acid and its derivatives (the major constituents of Boswellin) have anti-carcinogenic, anti-tumor, and anti-hyperlipidemic activities. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)