| In vivo: |
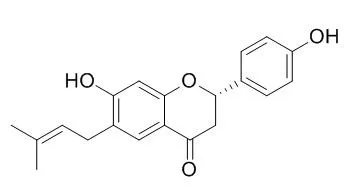
| Int J Mol Sci. 2016 Apr 8;17(4):527. | | Bavachin from Psoralea corylifolia Improves Insulin-Dependent Glucose Uptake through Insulin Signaling and AMPK Activation in 3T3-L1 Adipocytes.[Pubmed: 27070585] | The fruit of Psoralea corylifolia L. (Fabaceae) (PC), known as "Bo-Gol-Zhee" in Korea has been used as traditional medicine. Ethanol and aqueous extracts of PC have an anti-hyperglycemic effect by increasing plasma insulin levels and decreasing blood glucose and total plasma cholesterol levels in type 2 diabetic rats.
METHODS AND RESULTS:
In this study, we purified six compounds from PC and investigated their anti-diabetic effect. Among the purified compounds, Bavachin most potently accumulated lipids during adipocyte differentiation. Intracellular lipid accumulation was measured by Oil Red-O (ORO) cell staining to investigate the effect of compounds on adipogenesis. Consistently, Bavachin activated gene expression of adipogenic transcriptional factors, proliferator-activated receptorγ (PPARγ) and CCAAT/enhancer binding protein-α (C/EBPα). Bavachin also increased adiponectin expression and secretion in adipocytes. Moreover, Bavachin increased insulin-induced glucose uptake by differentiated adipocytes and myoblasts. In differentiated adipocytes, we found that Bavachin enhanced glucose uptake via glucose transporter 4 (GLUT4) translocation by activating the Akt and 5'AMP-activated protein kinase (AMPK) pathway in the presence or absence of insulin.
CONCLUSIONS:
These results suggest that Bavachin from Psoralea corylifolia might have therapeutic potential for type 2 diabetes by activating insulin signaling pathways. | | Journal of Emergency in Traditional Chinese Medicine, 2014, 23(9):1585-8. | | Effect of Brain Injury and Bavachin on 5-HT and VEGF of Rats with Tibial Fracture.[Reference: WebLink] | To observe the influence of brain injury and Bavachin on 5-HT and VEGF during the healing of rats′ tibial fracture.
METHODS AND RESULTS:
42 healthy male SD rats were selected and randomly divided into 7groups.For normal control group,blood was drawn on the third day.For the other groups,blood was drawn on postoperative day 3,7,14,21,28,and 35.And then centrifuged supernatant was acquired to determine the amount of VEGF and 5-HT by ELISA.(1) 5-TH:The concentration in the brain injury group and facture +brain injury group peaked in the first week,before it gradually declined in the following week.It reached a plateau 3 weeks later.Comparisons between the three fracture + Bavachin groups(all three doses) and the fracture group in one and two weeks have shown no significance(P 0.05).(2)VEGF:The concentrations in all treatment groups were higher than those in the control group.The concentration in fracture + brain injury group was higher than that in fracture group or brain injury group.After a week,the concentration of VEGF was in direct proportion to those of the Bavachin in the 3 facture + Bavachin groups.5 weeks later,they headed to similar concentration.
CONCLUSIONS:
The brain injury can induce increased VEGF content in peripheral blood to promote fracture healing.Bavachin in the blood can stimulate the genetic expression of VEGF in PB,and directly help the fracture healing.Also,the high does and the middle dose have better healing efficacy.At the beginning of brain injury,5-HT increases and is released into the cerebrum capillaries,accelerating the healing of fracture.However later 5-HT goes through the blood cerebrospinal fluid barrier and its concentration in the nervous centralis declines,resulting in the slowdown of fracture healing.Bavachin has no significant influence on the 5-HT concentration during the fracture healing. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)