| Description: |
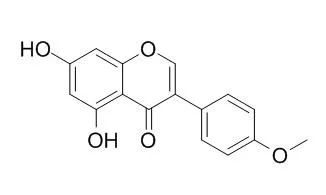
Biochanin A, an O-methylated natural isoflavonoid classified as phytoestrogen, is a naturally occurring fatty acid amide hydrolase (FAAH) inhibitor, which inhibits FAAH with IC50s of 1.8, 1.4 and 2.4 μM for mouse, rat, and human FAAH, respectively. Biochanin A has hypoglycemic, antilipemic,anti-tumorigenesis, anti-oxidation, and anti-inflammatory properties, it also has neuroprotective effects in cerebral ischemia/reperfusion by inhibiting inflammatory response and the inactivation of p38 signaling pathway. Biochanin A could inhibit Methicillin-resistant Staphylococcus aureus efflux system through reducing pathogen' s expression of nor A and norA protein. |
| Targets: |
TNF-α | IL Receptor | p38MAPK | LDL | Bcl-2/Bax | P-gp | EGFR | NO | NOS | IkB | NF-kB | IKK | FAAH |
| In vitro: |
| Wei Sheng Wu Xue Bao. 2014 Oct 4;54(10):1204-11. | | [Inhibitory effects of biochanin A on the efflux pump of methicillin-resistant Staphylococcus aureus (MRSA)].[Pubmed: 25803898] |
METHODS AND RESULTS:
To study the inhibitory effect of Biochanin A on efflux system of Methicillin-resistant Staphylococcus aureus (MRSA). Biochanin A alone had no inhibitory effect on MRSA41577, but it showed synergy effect with ciprofloxacin in inhibition MRSA41577 in which 40pg/mL Biochanin A decreased the minimum inhibitory concentration (MIC) value of ciprofloxacin from 64 microg/mL to 8 microgg/mL. Biochanin A significantly increased the accumulation of ciprofloxacin in MRSA41577 in a time-dependent manner. At 15 min, Biochanin A increased ciprofloxacin in MRSA41577 by 83%, which is similar to that of reserpine (positive control). Further mechanism studies indicated that Biochanin A could reduce the expression of nor A in ciprofloxacin-treated MRSA41577. After incubated with Biochanin A and ciprofloxacin for 16 h, the relative expression of nor A of MRSA41577 was reduced by 65%. SDS-PAGE analysis showed that the total protein profiles of MRSA41577 were significantly changed after treatment with Biochanin A for 16h, in which both norA protein and efflux system ABC transporter ATP-binding protein were significantly decreased.
CONCLUSIONS:
Biochanin A could inhibit Methicillin-resistant Staphylococcus aureus efflux system through reducing pathogen' s expression of nor A and norA protein. | | Eur J Pharmacol. 2011 Feb 25;653(1-3):8-15. | | Biochanin-A, an isoflavon, showed anti-proliferative and anti-inflammatory activities through the inhibition of iNOS expression, p38-MAPK and ATF-2 phosphorylation and blocking NFκB nuclear translocation.[Pubmed: 21147093 ] | Biochanin A, an isoflavone, existing in red clover, cabbage and alfalfa, has an inhibitory and apoptogenic effect on certain cancer cells. However, the actual mechanism by which this compound inhibits proliferation and induces apoptosis in cancer cells and the mechanism of its anti-inflammatory activities have not been well characterized. In this study, we have investigated the anti-inflammatory and anti-proliferative activity of Biochanin A.
METHODS AND RESULTS:
The effects of Biochanin A on RAW 264.7, HT-29 cell lines and mouse peritoneal macrophages have been investigated in vitro. Cell proliferation and anti-inflammatory effects were analyzed by 3-(4-5-dimethylthiozol-2-yl)2-5-diphenyl-tetrazolium bromide (MTT) assay, (3)H-thymidine incorporation assay, Western blot, cytokines estimation, Luciferase assay, Electrophoretic mobility shift assay (EMSA) and Kinase assay. Present investigation demonstrated that, Biochanin A inhibited lipopolysacharide (LPS)-induced nitric oxide(NO) production in macrophage and showed dose dependent inhibition of inducible nitric oxide synthase (iNOS) expression. The induction of NF-κB binding activity by LPS was inhibited markedly by co-incubation with different doses of Biochanin A. Biochanin A inhibited the LPS-induced IkB kinase (IKK) activity and nuclear factor kappa beta (NF-κB) activation associated with the inhibition of iNOS expression. LPS-induced phosphorylation of IκBα and p38 MAPK was blocked by Biochanin A and it inhibited IL-6, IL-1β and TNF-α production in RAW264.7 cells indicating its anti-inflammatory activity in association with anti-proliferation.
CONCLUSIONS:
Biochanin A is important for the prevention of phosphorylation and degradation of IκBα, thereby blocking NF-κB activation, which in turn leads to decreased expression of the iNOS, thus preventing proliferation and inflammation. |
|
| In vivo: |
| Iran Red Crescent Med J. 2014 Sep 5;16(9):e15424. | | Effect of biochanin a on serum visfatin level of streptozocin-induced diabetic rats.[Pubmed: 25593725] | Bioflavonoids are well known for their multi directional biologic activity including antidiabetic effect. It has been demonstrated that flavonoids can act as insulin secretagogue or insulin mimetic agents.
This experimental study was designed in Arak University of Medical Sciences, Arak, Iran, to investigate the effects of Biochanin A (a bioflavonoid) on fasting blood glucose (FBG), body weight, glycosylated hemoglobin (HbA1c), lipid profile, serum enzymes, and visfatin of streptozocin-induced diabetic rats.
METHODS AND RESULTS:
We used 24 male Wistar rats and randomly allocated them to four groups of six rats. One group was randomly assigned as control and diabetes was induced in three other groups by administration of streptozocin (35 mg/kg of body weight) intraperitoneally. The groups received the following treatments: group 1 (control), 5% DMSO; group 2 (diabetic control), 0.5% DMSO; and group 3 and 4, respectively 10 and 15 mg/kg Biochanin A for 30 days. Body weight and biochemical parameters including FBG, HbA1c, lipid profile, aspartate aminotransferase (AST), alanine aminotransferase (ALT), alkaline phosphatase (ALP), and visfatin were measured in all rats.
FBG level was significantly reduced in treated diabetic rats (139.8 ± 9.3 and 206 ± 11 mg/dL in groups 3 and 4, respectively) in comparison to the diabetic control (295.1 ± 14 mg/dL) (P < 0.05). Administration of Biochanin A significantly decreased HbA1c in group 3 (6.66 ± 0.33) and group 4 (7.11 ± 0.31) in comparison to the diabetic control group (8.26 ± 0.44) (P < 0.05). Levels of serum visfatin were improved to near normal levels in the treated rats (249 ± 35.5 and 161.33 ± 13.07 in groups 3 and 4, respectively) in comparison to the diabetic control (302.17 ± 19.4) (P < 0.05). Furthermore, Biochanin A showed a protective effect against weight loss in diabetic rats (P < 0.05). In treated rats, serum total cholesterol, triglyceride, and low-density lipoprotein cholesterol (LDL-c) were significantly decreased and high-density lipoprotein (HDL-c) was increased in comparison with the diabetic control group. In addition, Biochanin A restored the altered plasma enzymes (AST, ALT, and ALP) activities to near normal. Histopathologic examination of the pancreas also indicated that Biochanin A had protective effects on β-cells in streptozocin-induced diabetic rats.
CONCLUSIONS:
This study demonstrated that Biochanin A possessed hypoglycemic and antilipemic activities and could increase visfatin expression, which suggests its beneficial effect in the treatment of diabetes. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)