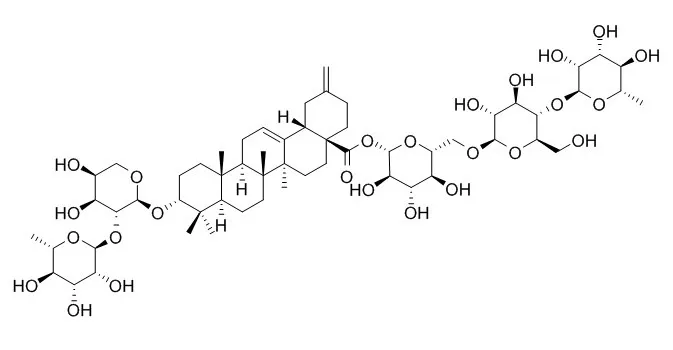
| Description: |
Ciwujianoside B can relieve fatigue, enhance memory, and improve human cognition, it may enhance reuse of choline to accelerate the synthesis of acetylcholine in hippocampal neurons by activation of cholinesterase. Ciwujianoside B has inhibitory effects on apoptosis induced by MIRI and cardiomyocytes apoptosis induced by H 2 O 2 in rats; it also shows radioprotective effects on the hematopoietic system in mice, which is associated with changes in the cell cycle, a reduction in DNA damage, and down-regulation of the ratio of Bax/Bcl-2 in bone marrow cells exposed to radiation. |
| Targets: |
P450 (e.g. CYP17) | AChR | NF-kB | Bcl-2/Bax |
| In vitro: |
| Food Chem. 2013 Dec 1;141(3):2426-33. | | Development of sample preparation method for Ciwujianoside B and E analysis in Acanthopanax senticosus by ionic liquids-ultrasound based extraction and high-performance liquid chromatography detection.[Pubmed: 23870977] | An ionic liquids-based ultrasonic-assisted extraction (ILUAE) method was successfully developed for extracting eleutheroside B and E from Radix Acanthopanax senticosus.
METHODS AND RESULTS:
Thirteen 1-alkyl-3-methylimidazolium ionic liquids with different cations and anions were investigated and 1-butyl-3-methylimidazolium bromide ([C4mim]Br) solution was selected as the solvent. The conditions for ILUAE, including the ionic liquid concentration, soaking time, ultrasonic power, ultrasonic time, solid-liquid ratio and number of extraction cycles, were optimized. With the proposed method, the energy consumption time was reduced to 30 min, whereas conventional method requires about 4h. The proposed method had good recovery (97.96-103.39%) and reproducibility (RSD, n=5; 3.3% for eleutheroside B, 4.6% for eleutheroside E).
CONCLUSIONS:
ILUAE was an efficient, rapid and simple sample preparation technique that showed high reproducibility and was environmental friendly. |
|
| In vivo: |
| Neural Regen Res. 2013 Apr 25;8(12):1103-12. | | Ciwujianoside B or E enhances learning and memory in experimentally aged rats.[Pubmed: 25206404] | Eleutheroside B or E, the main component of Acanthopanax, can relieve fatigue, enhance memory, and improve human cognition. Numerous studies have confirmed that high doses of acetylcholine significantly attenuate clinical symptoms and delay the progression of Alzheimer's disease.
METHODS AND RESULTS:
The present study replicated a rat model of aging induced by injecting quinolinic acid into the hippocampal CA1 region. These rats were intraperitoneally injected with low, medium and high doses of eleutheroside B or E (50, 100, 200 mg/kg), and rats injected with Huperzine A or PBS were used as controls. At 4 weeks after administration, behavioral tests showed that the escape latencies and errors in searching for the platform in a Morris water maze were dose-dependently reduced in rats treated with medium and high-dose eleutheroside B or E. Hematoxylin-eosin staining showed that the number of surviving hippocampal neurons was greater and pathological injury was milder in three eleutheroside B or E groups compared with model group. Hippocampal homogenates showed enhanced cholinesterase activity, and dose-dependent increases in acetylcholine content and decreases in choline content following eleutheroside B or E treatment, similar to those seen in the Huperzine A group.
CONCLUSIONS:
These findings indicate that eleutheroside B or E improves learning and memory in aged rats. These effects of eleutheroside B or E may be mediated by activation of cholinesterase or enhanced reuse of choline to accelerate the synthesis of acetylcholine in hippocampal neurons. | | Nutrients . 2019 May 22;11(5):1142. | | Memory Enhancement by Oral Administration of Extract of Eleutherococcus senticosus Leaves and Active Compounds Transferred in the Brain[Pubmed: 31121888] | | Abstract
The pharmacological properties of Eleutherococcus senticosus leaf have not been clarified although it is taken as a food item. In this study, the effects of water extract of Eleutherococcus senticosus leaves on memory function were investigated in normal mice. Oral administration of the extract for 17 days significantly enhanced object recognition memory. Compounds absorbed in blood and the brain after oral administration of the leaf extract were detected by LC-MS/MS analyses. Primarily detected compounds in plasma and the cerebral cortex were ciwujianoside C3, eleutheroside M, Ciwujianoside B, and ciwujianoside A1. Pure compounds except for ciwujianoside A1 were administered orally for 17 days to normal mice. Ciwujianoside C3, eleutheroside M, and Ciwujianoside B significantly enhanced object recognition memory. These results demonstrated that oral administration of the leaf extract of E. senticosus enhances memory function, and that active ingredients in the extract, such as ciwujianoside C3, eleutheroside M, and Ciwujianoside B, were able to penetrate and work in the brain. Those three compounds as well as the leaf extract had dendrite extension activity against primary cultured cortical neurons. The effect might relate to memory enhancement.
Keywords: Eleutheococcus senticosus leaf; blood-brain barrier; dendrite; memory; mice. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)