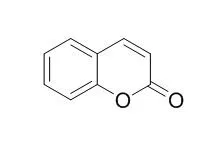
| In vitro: |
| J Photochem Photobiol B. 2015 May;146:44-51. | | Photoprotective effect of coumarin and 3-hydroxycoumarin in sea urchin gametes and embryonic cells.[Pubmed: 25795999] | Ultraviolet radiation B (UVB) represents 5% of all solar UV radiation and chronic exposure can induce harmful biological responses, including skin cancer. Prospection of new drugs with photoprotective properties and less toxic effects is constant and natural products have been the main options in this field. Coumarins are a group of natural phenolic compounds that shows several pharmacological activities.
METHODS AND RESULTS:
The aim of present work was to investigate the effect of Coumarin and six derivatives in sea urchin gametes and zygotes exposed to UVB. Embryonic development assay was used to monitor UVB embryotoxicity. Firstly, we demonstrated that Coumarin inhibited first embryonic cell division from 5 μM (EC50 = 52.9 μM) and its derivatives showed an embryotoxic effect ten times higher. Then, gametes or zygotes were treated with Coumarin compounds before or after UVB exposure (UVB doses ranged from 0.056 to 0.9 kJm(-2)). Pretreatment of gametes or zygotes with Coumarin or 3-hydroxyCoumarin (1 μM, both) decreased UVB embryotoxic effect. Protective effect of the compounds was observed only when cells were treated previous to UVB exposure. Coumarin derivatives 4-hydroxyCoumarin, 6-hydroxyCoumarin, 7-hydroxyCoumarin, 6,7-dihydroxyCoumarin and 6-methoxy-7-hydroxyCoumarin did not exhibit photoprotective activity.
CONCLUSIONS:
Our data provides evidences that Coumarin and 3-hydroxyCoumarin can be a promising class of photoprotective drugs. |
|
| In vivo: |
| Cancer Res. 2000 Feb 15;60(4):957-69. | | Chemoprevention of aflatoxin B1 hepatocarcinogenesis by coumarin, a natural benzopyrone that is a potent inducer of aflatoxin B1-aldehyde reductase, the glutathione S-transferase A5 and P1 subunits, and NAD(P)H:quinone oxidoreductase in rat liver.[Pubmed: 10706111] | Structurally diverse compounds can confer resistance to aflatoxin B1 (AFB1) hepatocarcinogenesis in the rat.
METHODS AND RESULTS:
Treatment with either phytochemicals [benzyl isothiocyanate, Coumarin (CMRN), or indole-3-carbinol] or synthetic antioxidants and other drugs (butylated hydroxyanisole, diethyl maleate, ethoxyquin, beta-naphthoflavone, oltipraz, phenobarbital, or trans-stilbene oxide) has been found to increase hepatic aldo-keto reductase activity toward AFB1-dialdehyde and glutathione S-transferase (GST) activity toward AFB1-8,9-epoxide in both male and female rats. Under the conditions used, the natural benzopyrone CMRN was a major inducer of the AFB1 aldehyde reductase (AFAR) and the aflatoxin-conjugating class-alpha GST A5 subunit in rat liver, causing elevations of between 25- and 35-fold in hepatic levels of these proteins. Induction was not limited to AFAR and GSTA5: treatment with CMRN caused similar increases in the amount of the class-pi GST P1 subunit and NAD(P)H: quinone oxidoreductase in rat liver. Immunohistochemistry demonstrated that the overexpression of AFAR, GSTA5, GSTP1, and NAD(P)H:quinone oxidoreductase affected by CMRN is restricted to the centrilobular (periacinar) zone of the lobule, sometimes extending almost as far as the portal tract. This pattern of induction was also observed with ethoxyquin, oltipraz, and trans-stilbene oxide. By contrast, induction of these proteins by beta-naphthoflavone and diethyl maleate was predominantly periportal. Northern blotting showed that induction of these phase II drug-metabolizing enzymes by CMRN was accompanied by similar increases in the levels of their mRNAs. To assess the biological significance of enzyme induction by dietary CMRN, two intervention studies were performed in which the ability of the benzopyrone to inhibit either AFB1-initiated preneoplastic nodules (at 13 weeks) or AFB1-initiated liver tumors (at 50 weeks) was investigated. Animals pretreated with CMRN for 2 weeks prior to administration of AFB1, and with continued treatment during exposure to the carcinogen for a further 11 weeks, were protected completely from development of hepatic preneoplastic lesions by 13 weeks. In the longer-term dietary intervention, treatment with CMRN before and during exposure to AFB1 for a total of 24 weeks was found to significantly inhibit the number and size of tumors that subsequently developed by 50 weeks.
CONCLUSIONS:
These data suggest that consumption of a CMRN-containing diet provides substantial protection against the initiation of AFB1 hepatocarcinogenesis in the rat. | | Stroke. 2014 Nov;45(11):3436-9. | | Use of coumarin anticoagulants and cerebral microbleeds in the general population.[Pubmed: 25316276] |
It remains undetermined whether the use of Coumarin anticoagulants associates with cerebral microbleeds in the general population. We investigated whether (1) Coumarin use relates to higher prevalence and incidence of microbleeds, (2) microbleeds are more frequent in people with higher maximum international normalized ratios (INRs), and (3) among Coumarin users, variability in INR associates with microbleed presence.
METHODS AND RESULTS:
From the population-based Rotterdam Study, 4945 participants aged ≥45 years were included in the cross-sectional analysis, and 3069 participants had follow-up brain MRI. Information on Coumarin use was obtained from automated pharmacy records. Coumarin users were monitored, and INR values were measured in consecutive visits. Presence and location of microbleeds were rated on brain MRI. We investigated the association of Coumarin use with microbleeds using multivariable logistic regression.
Overall, 8.6% had used Coumarin anticoagulants before the first MRI and 5.9% before follow-up MRI. The prevalence of microbleeds was 19.4%, and the incidence was 6.9% during a mean follow-up of 3.9 years (SD, 0.5). Compared with never users, Coumarin users had a higher prevalence of deep or infratentorial microbleeds and a higher incidence of any microbleeds, although statistical significance was not reached in the latter. A higher maximum INR was associated with deep or infratentorial microbleeds. Among Coumarin users, a greater variability in INR was associated with a higher prevalence of microbleeds.
CONCLUSIONS:
Coumarin use is associated with microbleeds. Associations were strongest for people with greater variability in INR. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)