| In vitro: |
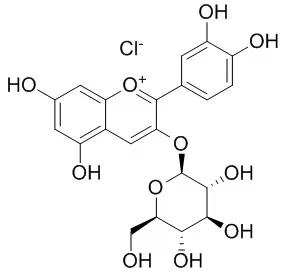
| Life Sci. 2003 Jul 18;73(9):1097-114. | | Protective effects of cyanidin-3-O-glucoside from blackberry extract against peroxynitrite-induced endothelial dysfunction and vascular failure.[Pubmed: 12818719] | Anthocyanins are a group of naturally occurring phenolic compounds as colorants in several plants, flowers and fruits. These pigments have a great importance as quality indicators, as chemotaxonomic markers and antioxidants.
METHODS AND RESULTS:
The content of blackberry (Rubus species) juice was investigated by HPLC/ESI/MS using narrow bore HPLC columns. Using this method we demonstrated that cyanidin-3-O-glucoside represents about 80% of the total anthocyanin contents in blackberry extract. Here we investigated antioxidant activity of the blackberry juice and cyanidin-3-O-glucoside on the endothelial dysfunction in cells and in vascular rings exposed to peroxynitrite. In human umbilical vein endothelial cells (HUVEC) in vitro, peroxynitrite caused a significant suppression of mitochondrial respiration (38 +/- 2.1% of control cells), as measured by the mitochondrial-dependent conversion of the dye MTT to formazan. Peroxynitrite caused DNA strand breakage (63 +/- 1.9% single strand vs 3 +/- 0.9% single strand in control cells), as measured by the alkaline unwinding assay, and caused an activation of PARS, as measured by the incorporation of radiolabeled NAD(+) to nuclear proteins. Blackberry juice (different dilutions that contained 80 ppm;40 ppm;14.5 ppm of cyanidin-3-O-glucoside) and cyanidin-3-O-glucoside (as chloride) (0.085 microM; 0.028 microM; 0.0085 microM) reduced the peroxynitrite-induced suppression of mitochondrial respiration, DNA damage and PARS activation in HUVECs. Vascular rings exposed to peroxynitrite exhibited reduced endothelium-dependent relaxant responses in response to acetylcholine as well as a vascular contractility dysfunction in response to norepinephrine. The development of this peroxynitrite-induced vascular dysfunction was ameliorated by the blackberry juice (different dilutions that contained 80 ppm;40 ppm;14.5 ppm of cyanidin-3-O-glucoside) and cyanidin-3-O-glucoside (as Cyanidin-3-O-glucoside chloride) (0.085 microM;0.028 microM;0.0085 microM).
CONCLUSIONS:
In conclusion our findings clearly demonstrate that blackberry juice containing cyanidin-3-O-glucoside is a scavenger of peroxynitrite and that exert a protective effect against endothelial dysfunction and vascular failure induced by peroxynitrite. | | Int J Mol Sci. 2016 Jul; 17(7): 993. | | Cytotoxicity and Apoptotic Effects of Polyphenols from Sugar Beet Molasses on Colon Carcinoma Cells in Vitro[Pubmed: 27347927] | | Three polyphenols were isolated and purified from sugar beet molasses by ultrasonic-aid extraction and various chromatographic techniques, and their structures were elucidated by spectral analysis. Cytotoxicity and the molecular mechanism were measured by methyl thiazolyl tetrazolium (MTT) assay, flow cytometry, caspase-3 activity assay and Western blot assay. The results showed that gallic acid, Cyanidin-3-O-glucoside chloride and epicatechin have cytotoxicity to the human colon, hepatocellular and breast cancer cells. Cyanidin-3-O-glucoside chloride showed its cytotoxicity against various tumor cell lines, particularly against colon cancer Caco-2 cells with half maximal inhibitory concentration (IC50) value of 23.21 ± 0.14 μg/mL in vitro. Cyanidin-3-O-glucoside chloride may be a potential candidate for the treatment of colon cancer. In the mechanism study, Cyanidin-3-O-glucoside chloride increased the ratio of cell cycle at G0/G1 phase and reduced cyclin D1 expression on Caco-2 cells. Cyanidin-3-O-glucoside chloride decreased mutant p21 expression, and increased the ratio of Bax/Bcl-2 and the activation of caspase-3 to induce apoptosis. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)