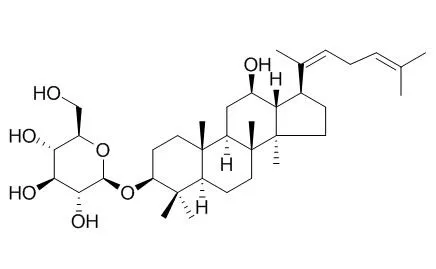
| Description: |
Ginsenoside Rh3 is a bacterial metabolite of Ginsenoside Rg5, Rh3 has anti-inflammatory effect in microglia by modulating AMPK and its downstream signaling pathways, it may improve chronic dermatitis or psoriasis by the regulation of IL-1β and TNF-α produced by macrophage cells and of IFN-γ produced by Th cells. Rg5 and Rh3 inhibited acetylcholinesterase activity in a dose-dependent manner, with IC50 values of 18.4 and 10.2 uM, respectively, they may protect memory deficit by inhibiting AChE activity and increasing BDNF expression and CREB activation. |
| Targets: |
COX | IFN-γ | IL Receptor | TNF-α | cAMP | PKC | NOS | HO-1 | PI3K | Akt | JAK | STAT | AMPK | NF-kB | Nrf2 |
| In vitro: |
| Int J Biochem Cell Biol. 1998 Mar;30(3):327-38. | | Ginsenoside Rh2 and Rh3 induce differentiation of HL-60 cells into granulocytes: modulation of protein kinase C isoforms during differentiation by ginsenoside Rh2.[Pubmed: 9611775] | Ginsenoside Rh3 and Rh4 were recently isolated from Panax ginseng, but their biochemical and pharmacological effects remain unidentified.
METHODS AND RESULTS:
The present study investigated whether the ginsenoside Rh group (G-Rh1, -Rh2, -Rh3 and -Rh4) having similar structures induce differentiation of HL-60 cells and whether protein kinase C (PKC) is involved in differentiation by ginsenoside. Differentiation was assessed by Wright-Giemsa stain and nitroblue tetrazolium reduction. G-Rh2 and Ginsenoside Rh3 induced differentiation of HL-60 cells into morphologically and functionally granulocytes but G-Rh1 and G-Rh4 did not. G-Rh2 and Ginsenoside Rh3 arrested the cell cycle at the G1/S phase, consistent with the ability to induce differentiation in a decreasing order of retinoic acid > G-Rh2 > Ginsenoside Rh3.
CONCLUSIONS:
It is concluded that G-Rh2 and Ginsenoside Rh3 can induce differentiation of HL-60 cells into granulocytes and modulation of PKC isoform levels may contribute to differentiation of HL-60 cells by G-Rh2. | | Free Radic Biol Med . 2018 Mar;117:238-246. | | Activation of Nrf2 by Ginsenoside Rh3 protects retinal pigment epithelium cells and retinal ganglion cells from UV[Pubmed: 29427790] | | Abstract
Excessive Ultra-violet (UV) radiation shall induce damages to resident retinal pigment epithelium (RPE) cells (RPEs) and retinal ganglion cells (RGCs). Here we tested the potential activity of Ginsenoside Rh3 ("Rh3") against the process. In cultured human RPEs and RGCs, pretreatment with Rh3 inhibited UV-induced reactive oxygen species (ROS) production and following apoptotic/non-apoptotic cell death. Rh3 treatment in retinal cells induced nuclear-factor-E2-related factor 2 (Nrf2) activation, which was evidenced by Nrf2 protein stabilization and its nuclear translocation, along with transcription of antioxidant responsive element (ARE)-dependent genes (HO1, NOQ1 and GCLC). Nrf2 knockdown by targeted-shRNA almost abolished Rh3-induced retinal cell protection against UV. Further studies found that Rh3 induced microRNA-141 ("miR-141") expression, causing downregulation of its targeted gene Keap1 in RPEs and RGCs. On the other hand, Rh3-induced Nrf2 activation and retinal cell protection were largely attenuated by the miR-141's inhibitor, antagomiR-141. In vivo, intravitreal injection of Rh3 inhibited retinal dysfunction by light damage in mice. Rh3 intravitreal injection also induced miR-141 expression, Keap1 downregulation and Nrf2 activation in mouse retinas. We conclude that Rh3 protects retinal cells from UV via activating Nrf2 signaling.
Keywords: Ginsenoside Rh3; MicroRNA-141; Nrf2, UV radiation; Oxidative stress; Retinal cells. |
|
| In vivo: |
| Arch Pharm Res. 2006 Aug;29(8):685-90. | | Inhibitory effect of ginsenoside Rg5 and its metabolite ginsenoside Rh3 in an oxazolone-induced mouse chronic dermatitis model.[Pubmed: 16964764] | The effect of a main constituent ginsenoside Rg5 isolated from red ginseng and its metabolite Ginsenoside Rh3 in a chronic dermatitis model was investigated.
METHODS AND RESULTS:
Ginsenosides Rg5 and Rh3 suppressed swelling of oxazolone-induced mouse ear contact dermatitis. These ginsenosides also reduced mRNA expressions of cyclooxygenase-2, interleukin (IL)-1beta, tumor necrosis factor (TNF)-alpha and interferon (IFN)-gamma. The inhibition of Ginsenoside Rh3 was more potent than that of ginsenoside Rg5.
CONCLUSIONS:
These findings suggest that Ginsenoside Rh3 metabolized from ginsenoside Rg5 may improve chronic dermatitis or psoriasis by the regulation of IL-1beta and TNF-alpha produced by macrophage cells and of IFN-gamma produced by Th cells. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)