| Description: |
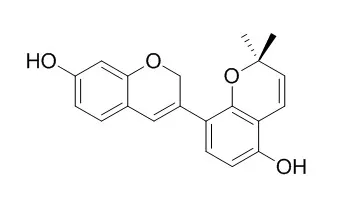
Glabrene, Liquiritin apioside, neolicuroside, and 18β-glycyrrhetic acid are the predominant phenolic derivatives partitioning at the interface and most likely the major contributors to the notable synergistic antioxidant activity when coupled with pea protein hydrolysates (PPHs).Glabrene has estrogen-like activity, it can stimulate DNA synthesis in human endothelial cells (ECV-304; E304) and has a bi-phasic effect on proliferation of human vascular smooth muscle cells (VSMC). Glabrene and isoliquiritigenin may serve as candidates for skin-lightening agents, they exert varying degrees of inhibition on tyrosinase-dependent melanin biosynthesis. |
| Targets: |
Tyrosinase | Estrogen receptor | Progestogen receptor |
| In vitro: |
| J Agric Food Chem. 2003 Feb 26;51(5):1201-7. | | Glabrene and isoliquiritigenin as tyrosinase inhibitors from licorice roots.[Pubmed: 12590456 ] | Tyrosinase is known to be a key enzyme in melanin biosynthesis, involved in determining the color of mammalian skin and hair. Various dermatological disorders, such as melasama, age spots, and sites of actinic damage, arise from the accumulation of an excessive level of epidermal pigmentation. The inadequacy of current therapies to treat these conditions as well as high cytotoxicity and mutagenicity, poor skin penetration, and low stability of formulations led us to seek new whitening agents to meet the medical requirements for depigmenting agents.
METHODS AND RESULTS:
The inhibitory effect of licorice extract on tyrosinase activity was higher than that expected from the level of glabridin in the extract. This led us to test for other components that may contribute to this strong inhibitory activity. Results indicated that Glabrene and isoliquiritigenin (2',4',4-trihydroxychalcone) in the licorice extract can inhibit both mono- and diphenolase tyrosinase activities. The IC(50) values for Glabrene and isoliquiritigenin were 3.5 and 8.1 microM, respectively, when tyrosine was used as substrate. The effects of Glabrene and isoliquiritigenin on tyrosinase activity were dose-dependent and correlated to their ability to inhibit melanin formation in melanocytes.
CONCLUSIONS:
This is the first study indicating that Glabrene and isoliquiritigenin exert varying degrees of inhibition on tyrosinase-dependent melanin biosynthesis, suggesting that isoflavenes and chalcones may serve as candidates for skin-lightening agents. |
|
| In vivo: |
| J Steroid Biochem Mol Biol. 2004 Jul;91(3):147-55. | | Estrogen-like activity of licorice root constituents: glabridin and glabrene, in vascular tissues in vitro and in vivo.[Pubmed: 15276622 ] | Post-menopausal women have higher incidence of heart diseases compared to pre-menopausal women, suggesting a protective role for estrogen. The recently Women's Health Initiative (WHI) randomized controlled trial concluded that the overall heart risk exceeded benefits from use of combined estrogen and progestin as hormone replacement therapy for an average of five years among healthy postmenopausal US women. Therefore, there is an urgent need for new agents with tissue-selective activity with no deleterious effects.
METHODS AND RESULTS:
In the present study, we tested the effects on vascular tissues in vitro and in vivo of two natural compounds derived from licorice root: glabridin, the major isoflavan, and Glabrene, an isoflavene, both demonstrated estrogen-like activities. Similar to estradiol-17beta (E2), glabridin (gla) stimulated DNA synthesis in human endothelial cells (ECV-304; E304) and had a bi-phasic effect on proliferation of human vascular smooth muscle cells (VSMC). Raloxifene inhibited gla as well as E2 activities. In animal studies, both intact females or after ovariectomy, gla similar to E2 stimulated the specific activity of creatine kinase (CK) in aorta (Ao) and in left ventricle of the heart (Lv). Glabrene (glb), on the other hand, had only the stimulatory effect on DNA synthesis in vascular cells, with no inhibition by raloxifene, suggesting a different mechanism of action. To further elucidate the mechanism of action of glb, cells were pre-incubated with glb and then exposed to either E2 or to gla; the DNA stimulation at low doses was unchanged but there was abolishment of the inhibition of VSMC cell proliferation at high doses as well as inhibition of CK stimulation by both E2 and by gla. We conclude that glb behaved differently than E2 or gla, but similarly to raloxifene, being a partial agonist/antagonist of E2. Glabridin, on the other hand, demonstrated only estrogenic activity.
CONCLUSIONS:
Therefore, we suggest the use of glb with or without E2 as a new agent for modulation of vascular injury and atherogenesis for the prevention of cardiovascular diseases in post-menopausal women. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)