| Description: |
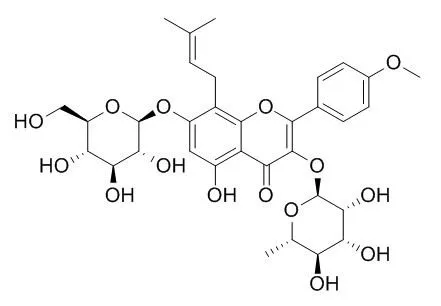
Icariin inhibits PDE5 and PDE4 activities with IC50s of 432 nM and 73.50 μM, respectively. Icariin also is a PPARα activator. Icariin has been reported to have anti-hypoxic, phytoestrogenic, anti-osteoporotic, anti-inflammatory, neuroprotective, and anti-depressant-like activities. Icariin is effective in the attenuation of AHR and chronic airway inflammatory changes in OVA-induced murine asthma model, and this effect is associated with regulation of Th17/Treg responses. Icariin inhibited NF-κB signaling activation and the NLRP3-inflammasome/caspase-1/IL-1β axis. |
| In vitro: |
| J Cell Biochem. 2015 Apr;116(4):580-8. | | Icariin inhibits foam cell formation by down-regulating the expression of CD36 and up-regulating the expression of SR-BI.[Pubmed: 25389062 ] | Icariin is an important pharmacologically active flavonol diglycoside that can inhibit inflammation in lipopolysaccharide (LPS)-stimulated macrophages. However, little is known about the molecular mechanisms underlying the inhibitory effect of Icariin in the formation of foam cells.
METHODS AND RESULTS:
In this study, macrophages were cultured with LPS and oxidized low-density lipoprotein (oxLDL) in the presence or absence of Icariin. RT-PCR and western blot were used to detect the levels of mRNA and protein expression of CD36, scavenger receptor class B type I (SR-BI) and the phosphorylation of p38MAPK. It was demonstrated that 4 μM or 20 μM Icariin treatment significantly inhibited the cholesterol ester (CE)/total cholesterol (TC) and oxLDL-mediated foam cell formation (P < 0.05). The binding of oxLDL to LPS-activated macrophages was also significantly hindered by Icariin (P < 0.05). Furthermore, Icariin down-regulated the expression of CD36 in LPS-activated macrophages in a dose-dependent manner and CD36 over-expression restored the inhibitory effect of Icariin on foam cell formation. The phosphorylation of p38MAPK was reduced by Icariin, indicating that Icariin reduced the expression of CD36 through the p38MAPK pathway. In addition, Icariin up-regulated SR-BI protein expression in a dose-dependent manner, and SR-BI gene silencing restored the inhibitory effect of Icariin on foam cell formation. These data demonstrate that Icariin inhibited foam cell formation by down-regulating the expression of CD36 and up-regulating the expression of SR-BI.
CONCLUSIONS:
Therefore, our findings provide a new explanation as to why Icariin could inhibit atherosclerosis. |
|
| In vivo: |
| Immunobiology. 2015 Jun;220(6):789-97. | | Regulation of Th17/Treg function contributes to the attenuation of chronic airway inflammation by icariin in ovalbumin-induced murine asthma model.[Pubmed: 25613226] | Icariin which is a flavonoid glucoside isolated from Epimedium brevicornu Maxim, has been reported to have anti-osteoporotic, anti-inflammatory and anti-depressant-like activities.
METHODS AND RESULTS:
In this study, we observed the effect of Icariin on airway inflammation of ovalbumin (OVA)-induced murine asthma model and the associated regulatory mode on T-helper (Th)17 and regulatory T (Treg) cell function. Our data revealed that chronic OVA inhalation induced a dramatic increase in airway resistance (RL) and decrease in the lung dynamic compliance (Cdyn), and Icariin and DEX treatment caused significant attenuation of such airway hyperresponsiveness (AHR). BALF cell counts demonstrated that Icariin and DEX led to a prominent reduction in total leukocyte as well as lymphocyte, eosinophil, neutrophil, basophil and monocyte counts. Histological analysis results indicated that Icariin and DEX alleviated the inflammatory cells infiltrating into the peribronchial tissues and goblet cells hyperplasia and mucus hyper-production. Flow cytometry test demonstrated that Icariin or DEX administration resulted in a significant percentage reduction in CD4+RORγt+ T cells and elevation of CD4+Foxp3+ T cells in BALF. Furthermore, Icariin or DEX caused a significant reduction in IL-6, IL-17 and TGF-β level in BALF. Unfortunately, Icariin had no effect on IL-10 level in BALF. Western blot assay found that Icariin or DEX suppressed RORγt and promoted Foxp3 expression in the lung tissue. qPCR analysis revealed that Icariin and DEX resulted in a notable decrease in RORγt and increase in Foxp3 mRNA expression in isolated spleen CD4+ T cell.
CONCLUSIONS:
In conclusion, our results suggested that Icariin was effective in the attenuation of AHR and chronic airway inflammatory changes in OVA-induced murine asthma model, and this effect was associated with regulation of Th17/Treg responses, which indicated that Icariin may be used as a potential therapeutic method to treat asthma with Th17/Treg imbalance phenotype. | | Int Immunopharmacol. 2016 Jan;30:157-162. | | Icariin attenuates cerebral ischemia-reperfusion injury through inhibition of inflammatory response mediated by NF-κB, PPARα and PPARγ in rats.[Pubmed: 26679678 ] | Icariin (ICA), an active flavonoid extracted from Chinese medicinal herb Epimedii, has been reported to exhibit many pharmacological effects including alleviating brain injury. However, little is known about the protection of ICA on ischemic stroke.
Hence, this study was designed to investigate the neuroprotective effect of ICA and explore its underlying mechanisms on ischemic stroke induced by cerebral ischemia-reperfusion (I/R) injury in rats.
METHODS AND RESULTS:
The animals were pretreated with ICA at doses of 10, 30mg/kg twice per day for 3 consecutive days followed by cerebral I/R injury induced by middle cerebral artery occlusion (MCAO) for 2h and reperfusion for 24h. Neurological function and infarct volume were observed at 24h after reperfusion, the protein expression levels of interleukin-1β (IL-1β), transforming growth factor-β1 (TGF-β1), PPARα and PPARγ, inhibitory κB-α (IκB-α) degradation and nuclear factor κB (NF-κB) p65 phosphorylation were detected by Western blot, respectively. It was found that pretreatment with ICA could decrease neurological deficit score, diminish the infarct volume, and reduce the protein levels of IL-1β and TGF-β1. Moreover, ICA suppressed IκB-α degradation and NF-κB activation induced by I/R. Furthermore, the present study also showed that ICA up-regulated PPARα and PPARγ protein levels.
CONCLUSIONS:
These findings suggest that ICA has neuroprotective effect on ischemic stroke in rats through inhibition of inflammatory responses mediated by NF-κB and PPARα and PPARγ. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)