| In vitro: |
| Phytochemistry. 2014 Jul;103:196-202. | | Diterpenoids and phenylethanoid glycosides from the roots of Clerodendrum bungei and their inhibitory effects against angiotensin converting enzyme and α-glucosidase.[Pubmed: 24726372 ] | Abietane derivatives, bungnates A, B, 15-dehydrocyrtophyllone A and 15-dehydro-17-hydroxycyrtophyllone A, and two phenylethanoid glycosides, bunginoside A and 3″,4″-di-O-acetylmartynoside, together with nine known abietane derivatives and fourteen known phenylethanoid glycosides, were isolated from dried roots of Clerodendrum bungei.
METHODS AND RESULTS:
Their structures were determined on the basis of detailed spectroscopic analyses and acidic hydrolysis. The absolute configuration of bunginoside A was established from analysis of CD data. Selected compounds were evaluated for inhibitory effects against angiotensin converting enzyme (ACE) and α-glucosidase.
CONCLUSIONS:
15-Dehydrocyrtophyllone A showed an ACE inhibitory effect, and verbascoside, Leucosceptoside A and isoacteoside exhibited strong inhibitory capacity against α-glucosidase. | | J Nat Prod. 1998 Nov;61(11):1410-2. | | Phenylethanoid glycosides from Digitalis purpurea and Penstemon linarioides with PKCalpha-inhibitory activity.[Pubmed: 9834166 ] | In a continuation of our search for potential tumor inhibitors from plants, it was found that the CH2Cl2-MeOH (1:1) extracts from Digitalis purpurea and Penstemon linarioides both showed PKCalpha-inhibitory bioactivity.
METHODS AND RESULTS:
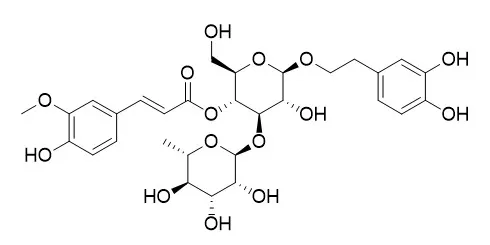
Bioassay-directed fractionation of the extract from D. purpurea yielded the new, weakly active phenylethanoid glycoside 2-(3-hydroxy-4-methoxy-phenyl)-ethyl-O-(alpha-L-rhamnosyl)-(1-->3) -O- (alpha-L-rhamnosyl)-(1-->6)-4-O-E-feruloyl-beta-D-glucopy ran oside (1) together with the four known compounds calceolarioside A (2), calceolarioside B (3), forsythiaside (4), and plantainoside D (5). The extract from P. linarioides yielded the three known glycosides Leucosceptoside A (6), acteoside (7), and poliumoside (8), together with the iridoid plantarenaloside (9).
CONCLUSIONS:
All of the isolated compounds, except compound 9, showed inhibitory activity against PKCalpha with IC50 values (in microM) of 125 (1), 0.6 (2), 4.6 (3), 1.9 (4), 14.8 (5), 19.0 (6), 9.3 (7), and 24.4 (8). |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)