| Description: |
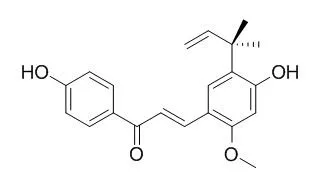
Licochalcone A is an estrogenic flavanoid extracted from licorice root, showing antimalarial, antileishmanial, anticancer, anti-inflammatory, antibacterial and antiviral activities. It could be a promising strategy in treating osteoporotic weight-bearing bones fractures with defects, and be a useful compound for the development of antibacterial agents for the preservation of foods containing high concentrations of salts and proteases, in which cationic peptides might be less effective.
|
| Targets: |
NF-kB | IL Receptor | IkB | P-gp | PARP | P450 (e.g. CYP17) | MMP(e.g.TIMP) | p65 | Caspase | TNF-α | NO | Antifection | IKK |
| In vitro: |
| Antimicrob Agents Chemother. 1994 Jul;38(7):1470-5. | | Licochalcone A, a new antimalarial agent, inhibits in vitro growth of the human malaria parasite Plasmodium falciparum and protects mice from P. yoelii infection.[Pubmed: 7979274] | Licochalcone A, isolated from Chinese licorice roots, inhibited the in vitro growth of both chloroquine-susceptible (3D7) and chloroquine-resistant (Dd2) Plasmodium falciparum strains in a [3H]hypoxanthine uptake assay. The growth inhibition of the chloroquine-resistant strain by Licochalcone A was similar to that of the chloroquine-susceptible strain.
METHODS AND RESULTS:
To examine the activity of Licochalcone A on the different asexual blood stages of the parasite, Licochalcone A was added to highly synchronized cultures containing rings, trophozoites, and schizonts. The growth of the parasites at all stages was inhibited by Licochalcone A. The in vivo activity of Licochalcone A was tested in a mouse model of infection with P. yoelii. Licochalcone A administered either intraperitoneally or orally for 3 to 6 days protected the mice from the otherwise lethal P. yoelii infection.
CONCLUSIONS:
These results demonstrate that Licochalcone A exhibits potent antimalarial activity and might be developed into a new antimalarial drug. | | Antimicrob Agents Chemother. 2002 May;46(5):1226-30. | | Antibacterial activity of licochalcone A against spore-forming bacteria.[Pubmed: 11959549 ] | Licochalcone A was isolated from the roots of licorice, Glycyrrhiza inflata, which has various uses in the food and pharmaceutical industries; isolation was followed by extraction with ethanol and column chromatography with silica gel.
METHODS AND RESULTS:
In this study, the activities of Licochalcone A against some food contaminant microorganisms were evaluated in vitro. The vegetative cell growth of Bacillus subtilis was inhibited in a Licochalcone A concentration-dependent manner and was completely prevented by 3 micrograms of Licochalcone A/ml. Licochalcone A showed a high level of resistance to heating at 80 to 121 degrees C for 15 min. Licochalcone A did not inhibit the germination of heat-treated spores of B. subtilis induced by L-alanine. Licochalcone A showed effects against all gram-positive bacteria tested and especially was effective against all Bacillus spp. tested, with MICs of 2 to 3 micrograms/ml, but was not effective against gram-negative bacteria or eukaryotes at 50 micrograms/ml. Although the cationic antimicrobial peptides protamine and epsilon-poly-L-lysine resulted in the loss of antimicrobial activity in the presence of either 3% (wt/vol) NaCl or protease at 20 micrograms/ml, the antibacterial activity of Licochalcone A was resistant to these conditions.
CONCLUSIONS:
Thus, Licochalcone A could be a useful compound for the development of antibacterial agents for the preservation of foods containing high concentrations of salts and proteases, in which cationic peptides might be less effective. |
|
| In vivo: |
| Exp Biol Med (Maywood). 2015 Jan;240(1):26-33. | | Role of licochalcone A on thymic stromal lymphopoietin expression: implications for asthma.[Pubmed: 25055998] | Asthma is a common chronic inflammatory disease characterized by the infiltration and accumulation of memory-like Th2 cells and eosinophils. Viral infection has emerged as the most common cause of severe episodes of asthma. For the treatment of bronchial asthma, the root of liquorice (Glycyrrhiza glabra) has been used as a traditional medicine in the East and West. Licochalcone A is the predominant, characteristic chalcone in liquorice root.
METHODS AND RESULTS:
To determine whether Licochalcone A possesses an anti-inflammatory effect, we tested its effect on the expression and production of thymic stromal lymphopoietin (TSLP) in BEAS 2B cells and primary bronchial epithelial cells. We found that polyinosinic-polycytidylic acid (poly-IC)-induced TSLP expression was suppressed by treatment with Licochalcone A in a dose- and time-dependent manner. We also found that poly-IC-induced mRNA expression of other proinflammatory mediators such as MCP-1, RANTES, and IL-8 was suppressed by Licochalcone A. Furthermore, Licochalcone A suppressed poly-IC-induced nuclear factor kappa B (NF-κB) nuclear translocation and DNA-binding activity by suppressing the Iκβ kinase (IKK) activity but not by direct phosphorylation of p65 at serine 276.
CONCLUSIONS:
Collectively, our findings suggest that Licochalcone A suppresses poly-IC-induced TSLP expression and production by inhibiting the IKK/NF-κB signaling pathway, which might be involved in the pathogenesis of virus-exacerbated asthma. Further elucidation of the mechanisms underlying these observations can help develop therapeutic strategies for virally induced asthma. | | Biomaterials. 2014 Mar;35(9):2789-97. | | The effect of licochalcone A on cell-aggregates ECM secretion and osteogenic differentiation during bone formation in metaphyseal defects in ovariectomized rats.[Pubmed: 24439395 ] | Treatment of weight-bearing bones fractures with defects is critical for patients with osteoporosis's rehabilitation. Although various tissue engineering methods were reported, the best treating strategy for tissue engineering remains to be identified as the limitation of enhancing the ability of the osteogenetic differentiation potential of seed cell is one of the cardinal issues to be solved. The objective of this study is to investigate the feasibility of applying licochalcone-A (L-A) and bone marrow mesenchymal stem cells (BMSC)-aggregate in bone repairing tissue engineering and further study the biological effects of L-A on the cell aggregate formation and osteogenic properties.
METHODS AND RESULTS:
80 female Sprague Dawley rats underwent bilateral ovariectomy were made with a 3.5 mm femurs bone defects without any fixation. These rats were then randomly assigned to five different treatment groups: (1) empty defect (n = 16), (2) CA-LA (n = 16), (3) CA-N (n = 16), (4) CA-L (n = 16), (5) CA-S (n = 16) and 16 female SD rats were treated as a control. Data showed that L-A administrated cell aggregate had a stronger osteogenic differentiation and mineralized formation potential than non-administrated group both in vitro and in vivo. For in vitro study, L-A administrated group had a more significant expression of ECM, osteogenic associated maker in addition with more mineralized area and higher ALP activity compared with the control group. For in vivo study, 3D reconstruction of micro-CT, HE staining and bone strength results showed that newly formed bone in groups administrated by L-A was significant higher than that in Sham group at 2, 4, 8 and 12 weeks after transplantation, especially for groups which was systematically injected with L-A at 8 weeks.
CONCLUSIONS:
Results of our study demonstrated that LA could positively affect cell behavior in cell-aggregate engineering and could be a promising strategy in treating osteoporotic weight-bearing bones fractures with defects. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)