Neuraminidase (NA), a key enzyme in viral replication, is the first-line drug target to combat influenza. On the basis of a shape-focused virtual screening, the roots of Glycyrrhiza glabra (licorice) were identified as plant species with an accumulation of constituents that show 3D similarities to known influenza NA inhibitors (NAIs).
METHODS AND RESULTS:
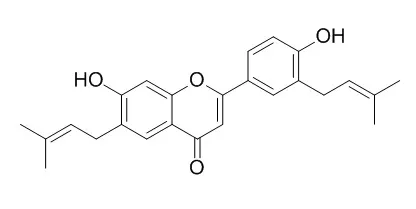
Phytochemical investigation revealed 12 constituents identified as (E)-1-[2,4-dihydroxy-3-(3-methyl-2-butenyl)phenyl]-3-(8-hydroxy-2,2-dimethyl-2H-1-benzopyran-6-yl)-2-propen-1-one (1), 3,4-dihydro-8,8-dimethyl-2H,8H-benzo[1,2-b:3,4-b']dipyran-3-ol (2), biochanin B (3), glabrol (4), glabrone (5), hispaglabridin B (6), Licoflavone B (7), licorice glycoside B (8), licorice glycoside E (9), liquiritigenin (10), liquiritin (11), and prunin (12). Eleven of these constituents showed significant influenza virus NA inhibition in a chemiluminescence (CL)-based assay. Additional tests, including (i) a cell-based cytopathic effect inhibition assay (general antiviral activity), (ii) the evaluation of cytotoxicity, (iii) the inhibition of the NA of Clostridium perfringens (CL- and fluorescence (FL)-based assay), and (iv) the determination of self-fluorescence and quenching, provided further perspective on their anti-influenza virus potential, revealing possible assay interference problems and false-positive results.
CONCLUSIONS:
Compounds 1, 3, 5, and 6 showed antiviral activity, most likely caused by the inhibition of NA. Of these, compounds 1, 3, and 6 were highly ranked in shape-focused virtual screening. |





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)