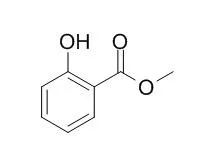
| Environ Microbiol. 2015 Apr;17(4):1365-76. |
| Plant methyl salicylate induces defense responses in the rhizobacterium Bacillus subtilis.[Pubmed: 25181478] |
Bacillus subtilis is a rhizobacterium that promotes plant growth and health. Cultivation of B. subtilis with an uprooted weed on solid medium produced pleat-like architectures on colonies near the plant. To test whether plants emit signals that affect B. subtilis colony morphology, we examined the effect of plant-related compounds on colony morphology.
METHODS AND RESULTS:
Bacillus subtilis formed mucoid colonies specifically in response to Methyl salicylate, which is a plant-defense signal released in response to pathogen infection. Methyl salicylate induced mucoid colony formation by stimulating poly-γ-glutamic acid biosynthesis, which formed enclosing capsules that protected the cells from exposure to antimicrobial compounds. Poly-γ-glutamic acid synthesis depended on the DegS-DegU two-component regulatory system, which activated DegSU-dependent gene transcription in response to Methyl salicylate. Bacillus subtilis did not induce plant Methyl salicylate production, indicating that the most probable source of Methyl salicylate in the rhizosphere is pathogen-infected plants. Methyl salicylate induced B. subtilis biosynthesis of the antibiotics bacilysin and fengycin, the latter of which exhibited inhibitory activity against the plant pathogenic fungus Fusarium oxysporum.
CONCLUSIONS:
We propose that B. subtilis may sense plants under pathogen attack via Methyl salicylate, and express defense responses that protect both B. subtilis and host plants in the rhizosphere. |
| Drug Test Anal. 2014 Jun;6 Suppl 1:67-73. |
| Hair analysis as a useful procedure for detection of vapour exposure to chemical warfare agents: simulation of sulphur mustard with methyl salicylate.[Pubmed: 24817050 ] |
Chemical warfare agents (CWA) are highly toxic compounds which have been produced to kill or hurt people during conflicts or terrorist attacks.
Despite the fact that their use is strictly prohibited according to international convention, populations' exposure still recently occurred. Development of markers of exposure to CWA is necessary to distinguish exposed victims from unexposed ones.
METHODS AND RESULTS:
We present the first study of hair usage as passive sampler to assess contamination by chemicals in vapour form. This work presents more particularly the hair adsorption capacity for Methyl salicylate used as a surrogate of the vesicant sulphur mustard. Chemical vapours toxicity through the respiratory route has historically been defined through Haber's law's concentration-time (Ct) product, and vapour exposure of hair to Methyl salicylate was conducted with various times or doses of exposure in the range of incapacitating and lethal Ct products corresponding to sulphur mustard. Following exposure, extraction of Methyl salicylate from hair was conducted by simple soaking in dichloromethane. Methyl salicylate could be detected on hair for vapour concentration corresponding to about one fifth of the sulphur mustard concentration that would kill 50% of exposed individuals (LCt50). The amount of Methyl salicylate recovered from hair increased with time or dose of exposure.
CONCLUSIONS:
It showed a good correlation with the concentration-time product, suggesting that hair could be used like a passive sampler to assess vapour exposure to chemical compounds. It introduces great perspectives concerning the use of hair as a marker of exposure to CWA. |
| J Am Mosq Control Assoc. 2014 Sep;30(3):199-203. |
| Fumigant Activity of 6 Selected Essential Oil Compounds and Combined Effect of Methyl Salicylate And Trans-Cinnamaldehyde Against Culex pipiens pallens.[Pubmed: 25843095] |
We studied the knockdown activity and lethal toxicity of 6 essential oil compounds-Methyl salicylate, linalool, 2-phenethyl alcohol, eugenol, β-citronellol, and trans-cinnamaldehyde-as fumigants against adult female Culex pipiens pallens in the laboratory.
METHODS AND RESULTS:
Of the 6 products tested, trans-cinnamaldehyde was the most toxic (LC50 = 0.26 μl/l air, 24 h) with a slow knockdown time (KT95 = 176.5 min at 0.5 μl/l air). Methyl salicylate displayed a lower toxicity (LC50 = 1.17 μl/l air, 24 h) but the fastest knockdown activity (KT95 = 16.8 min) at the sublethal concentration 0.5 μl/l air. Furthermore, the binary mixture of Methyl salicylate and trans-cinnamaldehyde exhibited a combined effect of fast knockdown activity and high toxicity against Cx. p. pallens adults, showing potential for development as natural fumigants for mosquito control. |





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)