| In vitro: |
| J Med Food. 2015 Apr;18(4):403-8. | | Neuroprotective effect of prenylated arylbenzofuran and flavonoids from morus alba fruits on glutamate-induced oxidative injury in HT22 hippocampal cells.[Pubmed: 25514545] | A prenylated arylbenzofuran and six flavonoids were isolated from the fruits of Morus alba L. through silica gel, octadecyl silica gel, and Diaion HP-20 column chromatography.
METHODS AND RESULTS:
Based on the nuclear magnetic resonance, mass spectrometry, and infrared spectroscopic data, the chemical structures of the compounds were determined to be artoindonesianin O (1), isobavachalcone (2), Morachalcone A (3), quercetin (4), astragalin (5), isoquercetin (6), and rutin (7). The isolated compounds were evaluated for protection of HT22-immortalized hippocampal cells against glutamate-induced oxidative stress.
CONCLUSIONS:
Compounds 1 and 3 exhibited protective effects with EC(50) values of 19.7±1.2 and 35.5±2.1 μM, respectively. The major compounds 1-3 and 7 were quantified using liquid chromatography/mass spectrometry analysis and were determined to be 1.88±2.1, 1.90±1.8, 0.78±1.5, and 37.29±2.2 mg/kg, respectively, in the ethanol extract of M. alba L. fruits. | | J Nat Prod. 2012 Nov 26;75(11):1951-5. | | Tyrosinase inhibitors from the wood of Artocarpus heterophyllus.[Pubmed: 23113717 ] |
METHODS AND RESULTS:
From the methanolic-soluble extract of the wood of Artocarpus heterophyllus, four new flavones, artocarmins A-D (1-4), and three new chalcones, artocarmitins A-C (5-7), have been isolated together with 13 known compounds. Their structures were determined on the basis of the spectroscopic data.
CONCLUSIONS:
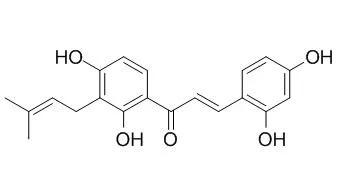
Compounds 1-4, 6, 7, 9-16, and 20 displayed significant tyrosinase inhibitory activity. The most active compound, Morachalcone A (12) (IC50, 0.013 μM), was 3000 times more active as a tyrosinase inhibitor than a positive control, kojic acid (IC50, 44.6 μM). |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)