Morus alba (white mulberry) has been used in traditional Chinese medicine as an anti-headache, diuretic, expectorant, and anti-diabetic agent. In previous studies, extracts of Morus alba demonstrated favorable biological properties, such as antioxidant activity, suppression of lipoxygenase (LOX)-1, cytotoxicity against cancer cells, and inhibition of the invasion and migration of cancer cells.
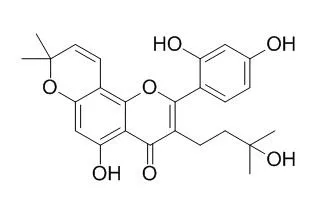
This study further evaluated the effects of Morusinol, a flavonoid derived from Morus alba root bark, on platelet aggregation and thromboxane B(2) (TXB(2) formation in vitro and thrombus formation in vivo.
METHODS AND RESULTS:
The antiplatelet potential of Morusinol was measured using in vitro rabbit platelet aggregation and TXB(2) formation assays. Arterial thrombus formation was investigated using an in vivo ferric chloride (FeCl(3)-induced thrombosis model.
Morusinol significantly inhibited collagen- and arachidonic acid-induced platelet aggregation and TXB(2) formation in cultured platelets in a concentration-dependent manner. Thrombus formation was reduced by 32.1, 42.0, and 99.0% for collagen-induced TXB(2) formation, and 8.0, 24.1, and 29.2% for arachadonic acid-induced TXB(2) formation, with 5, 10, and 30 μg/mL Morusinol, respectively. Moreover, oral Morusinol (20 mg/kg) or aspirin (20 mg/kg) for three days significantly increased the time to occlusion in vivo by 20.3±5.0 or 6.8±2.9 min, respectively, compared with the control (1% CMC, carboxymethyl cellulose).
CONCLUSIONS:
Taken together, these results indicate that Morusinol may significantly inhibit arterial thrombosis in vivo due to antiplatelet activity. Thus, Morusinol may exert beneficial effects on transient ischemic attacks or stroke via the modulation of platelet activation. |





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)