| In vitro: |
| Eur. J.Org. Chem. 2011, 2011(20-21): 3809-14. | | Protoberberine Alkaloids and Cancer Chemopreventive Properties of Compounds from Alangium salviifolium[Reference: WebLink] |
METHODS AND RESULTS:
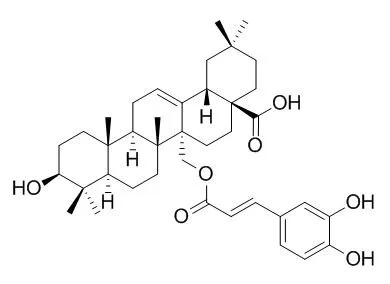
New protoberberine alkaloids, namely alangiumkaloids A (1) and B (2), 27-O-trans-caffeoylcylicodiscic acid (3), and β-d-glucopyranos-1-yl N-methylpyrrole-2-carboxylate (5) together with Myriceric acid B (4), isoalangiside (6), alangiside (7), 3-O-demethyl-2-O-methylalangiside (8), and demethylalangiside (9) have been isolated from Alangium salviifolium. The cancer chemopreventive properties and cytotoxic activities of the isolated compounds were evaluated.
CONCLUSIONS:
Compounds Myriceric acid B, 4, and 9 scavenged DPPH free radicals with IC50 values of 21.4, 21.8, and 24.0 μm, respectively. Alangisides 7 and 9 inhibited superoxide anion radical formation in the xanthine/xanthine oxidase (XXO) assay with IC50 values of 19.4 and 5.3 μm, respectively. Compounds 6–9 showed excellent antioxidant activity in the oxygen radical absorbance capacity (ORAC) assay with 12.8–24.9 ORAC units. Compounds 3 and Myriceric acid B inhibited aromatase activity with IC50 values of 4.7 and 6.8 μm, respectively. Although the isolated compounds showed only weak cytotoxicity or were inactive, compounds 3 and Myriceric acid B exhibited cytotoxic activity towards the MOLT-3 cell line with IC50 values of 5.6 and 3.9 μm, respectively. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)