| Kinase Assay: |
| Pharm Biol. 2014 Sep;52(9):1166-9. | | α-Glucosidase inhibition, 15-lipoxygenase inhibition, and brine shrimp toxicity of extracts and isolated compounds from Terminalia macroptera leaves.[Pubmed: 24635511] | Terminalia macroptera Guill. & Perr. (Combretaceae), a tree that grows in West Africa, has been used in traditional medicine against a variety of diseases such as hepatitis, gonorrhea, skin diseases, and diabetes. To investigate enzyme inhibitory activity against α-glucosidase and 15-lipoxygenase (15-LO) and toxicity against brine shrimp of extracts and compounds from T. macroptera leaves.
METHODS AND RESULTS:
Methanol extract, ethyl acetate, and butanol extracts obtained from the methanol extract, six isolated polyphenols (chebulagic acid, chebulic acid trimethyl ester, corilagin, methyl gallate, Narcissoside, and rutin), and shikimic acid were evaluated for enzyme inhibition and toxicity. In enzyme inhibition assays, all extracts showed high or very high activity. Chebulagic acid showed an IC50 value of 0.05 μM towards α-glucosidase and 24.9 ± 0.4 μM towards 15-LO, in contrast to positive controls (acarbose: IC50 201 ± 28 μM towards α-glucosidase, quercetin: 93 ± 3 μM towards 15-LO). Corilagin and Narcissoside were good 15-LO and α-glucosidase inhibitors, as well, while shikimic acid, methyl gallate, and chebulic acid trimethyl ester were less active or inactive. Rutin was a good α-glucosidase inhibitor (IC50 ca. 3 μM), but less active towards 15-LO. None of the extracts or the isolated compounds seemed to be very toxic in the brine shrimp assay compared with the positive control podophyllotoxin.
CONCLUSIONS:
Inhibition of α-glucosidase in the gastrointestinal tract may be a rationale for the medicinal use of T. macroptera leaves against diabetes in traditional medicine in Mali. The plant extracts and its constituents show strong inhibition of the peroxidative enzyme 15-LO. |
|
| Structure Identification: |
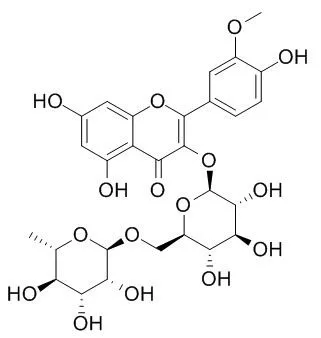
| J Sep Sci. 2014 Apr;37(8):957-65. | | Extraction and isolation of flavonoid glycosides from Flos Sophorae Immaturus using ultrasonic-assisted extraction followed by high-speed countercurrent chromatography.[Pubmed: 24515421] |
METHODS AND RESULTS:
A method of ultrasonic-assisted extraction followed by high-speed countercurrent chromatography was established for the extraction and isolation of three flavonoid glycosides, i.e. rutin, Narcissoside, and nicotiflorin from Flos Sophorae Immaturus. The effects of ultrasonic-assisted extraction factors for the main flavonoid compound (rutin) from Flos Sophorae Immaturus were optimized using Box-Behnken design combined with response surface methodology. The optimum conditions were determined as ultrasonic power 83% (600 W), solvent-to-material ratio 56:1, methanol concentration 82% v/v, and extraction time 60 min. Three bioactive flavonol glucosides, rutin, Narcissoside, and nicotiflorin were isolated from Flos Sophorae Immaturus using high-speed countercurrent chromatography. The separation was performed with a two-phase solvent system containing ethyl acetate/n-butanol/methanol/water (4:0.9:0.2:5, v/v). Amounts of 87 mg of rutin, 10.8 mg of Narcissoside, and 1.8 mg of nicotiflorin were isolated from 302 mg of crude extract of Flos Sophorae Immaturus in a one-step separation within 160 min with purities of 99.3, 98.0, and 95.1%, respectively, as determined by HPLC with diode array detection. Their structures were characterized by UV, MS, and NMR spectroscopy.
CONCLUSIONS:
It was demonstrated that the established method was simple, fast, and convenient, which was feasible to extract and isolate active flavonoid glycosides from Flos Sophorae Immaturus. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)