| Food Chem Toxicol. 2013 May;55:411-23. |
| Prunin- and hesperetin glucoside-alkyl (C4-C18) esters interaction with Jurkat cells plasma membrane: consequences on membrane physical properties and antioxidant capacity.[Pubmed: 23354392 ] |
Prunin (P)- and hesperetin glucoside (HG)-alkyl esters are lipid-soluble compounds with antimicrobial and antioxidant capacities in vitro.
METHODS AND RESULTS:
The effects of Prunin- and HG-alkyl (C4-C18) esters (0.1-100μM) on human leukemia T (Jurkat) cells viability and plasma membrane fluidity were evaluated. At this concentration, cell hyperpolarization and shrinkage were also observed. Cell plasma membrane fluidity was not affected, regardless the depths of the membrane level evaluated, but mild changes in plasma membrane hydration were found. Esterification did not affect the antioxidant capacity of Prunin and HG (0.1-10μM) against 1mM H2O2. When exposed to 1mM AAPH, P-alkyl esters retained Prunin antioxidant capacity, but HG-derivatives acted as pro-oxidants.
CONCLUSIONS:
Together, present experimental evidences suggest that short term exposures to 0.1-10μM concentrations of Prunin- and HG-alkyl (C4-C18) esters can be considered safe for cultured human cells, and further studies are required to investigate their long term effects, as well their safety for human consumption. |
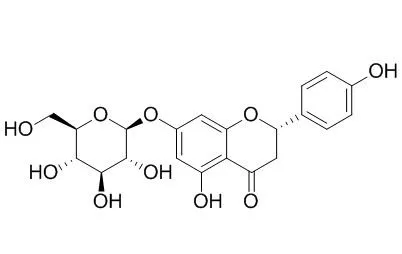
| Food Chemistry, 2011, 124(1):234-241. |
| Purification and characterisation of Aspergillus sojae naringinase: The production of prunin exhibiting markedly enhanced solubility with in vitro inhibition of HMG-CoA reductase.[Reference: WebLink] |
Aspergillus sojae isolated from a traditional Korean fermented soybean product exhibited strong naringinase activity.
METHODS AND RESULTS:
The naringinase enzyme was purified and had a molecular weight of 70kDa. The α-l-rhamnosidase activity of this enzyme was optimal at pH 6.0 and stable in the pH range of 5.5-8.0. The purified enzyme also had β-d-glucosidase activity, but the activity was relatively weak compared to the activity of the naringinase from Penicillium decumbens. The enzymatic bioconversion by A. sojae naringinase of naringin to Prunin was efficiently performed with a 91% yield and a negligible amount of naringenin. The bioconversion was achieved by repetitive batch reactions with enzyme recycling. Prunin exhibited a markedly enhanced solubility compared to naringenin and naringin while maintaining the in vitro inhibition of HMG-CoA reductase.
CONCLUSIONS:
The results reported in this paper show that the naringinase produced by A. sojae will be useful in enhancing the potential bioavailability of naringin by efficiently converting it to Prunin as a food component in citrus. |
| J Appl Microbiol. 2010 Oct;109(4):1450-7. |
| Antimicrobial properties of prunin, a citric flavanone glucoside, and its prunin 6″-O-lauroyl ester.[Pubmed: 20553344] |
To determine the antimicrobial potential of Prunin (P), a flavanone glucoside resulting from the hydrolysis of naringin present in grapefruit, and of its Prunin 6″-O-lauroyl ester (PL), synthesized by enzymatic catalysis.
METHODS AND RESULTS:
P and its lauroyl ester were tested against Gram-negative and Gram-positive bacteria, yeasts and moulds.Prunin showed no inhibitory effect against the micro-organisms assayed, but stimulated growth of Pseudomonas aeruginosa and different Bacilllus sp. A bactericidal effect was also determined on L. monocytogenes sensitive and bacteriocin-resistant mutant strains.Prunin did not show any antimicrobial property at all.
CONCLUSIONS:
PL is a potential antimicrobial compound with a high anti-Listeria property. |





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)