| Kinase Assay: |
| Journal of Medicinal Plant Research, 2009, 3(11):914-920. | | Compounds with elastase inhibition and free radical scavenging activities from Callistemon lanceolatus.[Reference: WebLink] |
METHODS AND RESULTS:
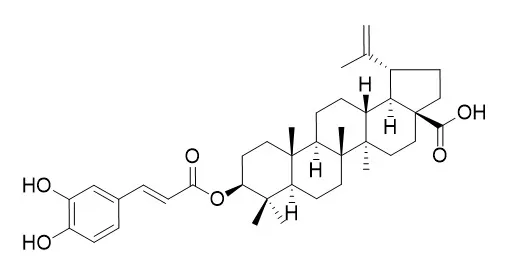
Bioassay-guided investigation of the stems of Callistemon lanceolatus led to the isolation of five compounds such as betulinic acid (1), Pyracrenic acid (2), arjunolic acid (3), catechin (4), and piceatannol (5). Their structures were elucidated on the basis of spectral studies as well as by comparison with literature data. Elastase inhibition and DPPH radical scavenging activities wereinvestigated for the isolated compounds.
CONCLUSIONS:
Among these, Pyracrenic acid (2) was identified as a potent elastase inhibitor with an IC50 value of 1.5 mg/mL, more active than the control compound, oleanolic acid (IC50 = 3.0 mg/mL). With regard to antioxidative studies, strong to moderate DPPH radical scavenging activities were observed with Pyracrenic acid (2), catechin (4) and piceatanol (5). |
|
| Structure Identification: |
| Zhong Yao Cai. 2012 Jun;35(6):904-8. | | Chemical constituents from the aerial roots of Ficus microcarpa.[Pubmed: 23236824] | To study the chemical constituents of the aerial roots of Ficus microcarpa.
METHODS AND RESULTS:
The compounds were isolated and purified by column chromatographic methods on silica gel and Sephadex LH-20. Their structures were elucidated by physicochemical properties and spectral data. RESULTS: 12 compounds were isolated from the 95% ethanol extract. Their structures were idenified as 3beta-hydroxy-11-oxours-12-ene (1), 3beta-acetoxy-11-oxours-12-ene (2), oleanic acid (3), 3beta-hydroxy-oleana-11,13 (18)-dien-28-oic acid (4), betulinic acid (5), Pyracrenic acid (6), platanic acid (7), isowigtheone (8), myrsininone A (9), derrone (10), alpinumisoflavone (11) and caffeic acid methyl ester (12), respectively.
CONCLUSIONS:
Compounds 4, 6, 12 are obtained from this genus for the first time, compounds 1, 7 - 11 are isolated from this plant for the first time. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)