| In vitro: |
| Oncogene. 2013 Oct;32(40):4825-35. | | Inhibition of the Nrf2 transcription factor by the alkaloid trigonelline renders pancreatic cancer cells more susceptible to apoptosis through decreased proteasomal gene expression and proteasome activity.[Pubmed: 23108405 ] | Evidence accumulates that the transcription factor nuclear factor E2-related factor 2 (Nrf2) has an essential role in cancer development and chemoresistance, thus pointing to its potential as an anticancer target and undermining its suitability in chemoprevention. Through the induction of cytoprotective and proteasomal genes, Nrf2 confers apoptosis protection in tumor cells, and inhibiting Nrf2 would therefore be an efficient strategy in anticancer therapy.
METHODS AND RESULTS:
In the present study, pancreatic carcinoma cell lines (Panc1, Colo357 and MiaPaca2) and H6c7 pancreatic duct cells were analyzed for the Nrf2-inhibitory effect of the coffee alkaloid Trigonelline (trig), as well as for its impact on Nrf2-dependent proteasome activity and resistance to tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) and anticancer drug-induced apoptosis. Chemoresistant Panc1 and Colo357 cells exhibit high constitutive Nrf2 activity, whereas chemosensitive MiaPaca2 and H6c7 cells display little basal but strong tert-butylhydroquinone (tBHQ)-inducible Nrf2 activity and drug resistance. Trig efficiently decreased basal and tBHQ-induced Nrf2 activity in all cell lines, an effect relying on a reduced nuclear accumulation of the Nrf2 protein. Along with Nrf2 inhibition, trig blocked the Nrf2-dependent expression of proteasomal genes (for example, s5a/psmd4 and α5/psma5) and reduced proteasome activity in all cell lines tested. These blocking effects were absent after treatment with Nrf2 siRNA, a condition in which proteasomal gene expression and proteasome activity were already decreased, whereas siRNA against the related transcription factor Nrf1 did not affect proteasome activity and the inhibitory effect of trig. Depending on both Nrf2 and proteasomal gene expression, the sensitivity of all cell lines to anticancer drugs and TRAIL-induced apoptosis was enhanced by trig. Moreover, greater antitumor responses toward anticancer drug treatment were observed in tumor-bearing mice when receiving trig.
CONCLUSIONS:
In conclusion, representing an efficient Nrf2 inhibitor capable of blocking Nrf2-dependent proteasome activity and thereby apoptosis protection in pancreatic cancer cells, trig might be beneficial in improving anticancer therapy. |
|
| In vivo: |
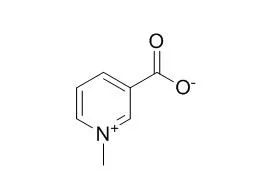
| Mol Nutr Food Res. 2014 Jul;58(7):1457-64. | | Unfavorable effect of trigonelline, an alkaloid present in coffee and fenugreek, on bone mechanical properties in estrogen-deficient rats.[Pubmed: 24867387] | Trigonelline (1-methylpyridinium-3-carboxylate), an alkaloid present in coffee and fenugreek seed, has been reported to exhibit phytoestrogenic activity. The aim of the present study was to investigate the effects of Trigonelline on bone mechanical properties of rats with normal estrogen level and estrogen deficiency (developing osteoporosis).
METHODS AND RESULTS:
The experiments were performed on 3-month-old nonovariectomized and ovariectomized (estrogen-deficient) Wistar rats, divided into control rats and rats receiving Trigonelline (50 mg/kg p.o. daily) for 4 weeks. The ovariectomy was performed 7-8 days before the start of Trigonelline administration. Serum bone turnover markers and bone mineralization, as well as mechanical properties of the tibial metaphysis, femoral diaphysis, and femoral neck were examined. Estrogen deficiency caused worsening of bone mineralization and mechanical properties of the tibial metaphysis, as well as increases in bone turnover markers. Administration of Trigonelline did not affect the investigated parameters in nonovariectomized rats, but it worsened the mineralization and mechanical properties of cancellous bone in ovariectomized rats.
CONCLUSIONS:
Unfavorable effects of Trigonelline on the skeletal system depended on the estrogen status. They were observed only in cancellous bone of estrogen-deficient rats. | | Diabetes Care. 2009 Jun;32(6):1023-5. | | Acute effects of decaffeinated coffee and the major coffee components chlorogenic acid and trigonelline on glucose tolerance.[Pubmed: 19324944 ] | Coffee consumption has been associated with lower risk of type 2 diabetes. We evaluated the acute effects of decaffeinated coffee and the major coffee components chlorogenic acid and Trigonelline on glucose tolerance.
METHODS AND RESULTS:
We conducted a randomized crossover trial of the effects of 12 g decaffeinated coffee, 1 g chlorogenic acid, 500 mg Trigonelline, and placebo (1 g mannitol) on glucose and insulin concentrations during a 2-h oral glucose tolerance test (OGTT) in 15 overweight men.
Chlorogenic acid and Trigonelline ingestion significantly reduced glucose (-0.7 mmol/l, P = 0.007, and -0.5 mmol/l, P = 0.024, respectively) and insulin (-73 pmol/l, P = 0.038, and -117 pmol/l, P = 0.007) concentrations 15 min following an OGTT compared with placebo. None of the treatments affected insulin or glucose area under the curve values during the OGTT compared with placebo.
CONCLUSIONS:
Chlorogenic acid and Trigonelline reduced early glucose and insulin responses during an OGTT. | | Nutrients . 2016 Mar 2;8(3):133. | | Effects of Trigonelline, an Alkaloid Present in Coffee, on Diabetes-Induced Disorders in the Rat Skeletal System[Pubmed: 26950142] | | Abstract
Diabetes increases bone fracture risk. Trigonelline, an alkaloid with potential antidiabetic activity, is present in considerable amounts in coffee. The aim of the study was to investigate the effects of Trigonelline on experimental diabetes-induced disorders in the rat skeletal system. Effects of Trigonelline (50 mg/kg p.o. daily for four weeks) were investigated in three-month-old female Wistar rats, which, two weeks before the start of Trigonelline administration, received streptozotocin (60 mg/kg i.p.) or streptozotocin after nicotinamide (230 mg/kg i.p.). Serum bone turnover markers, bone mineralization, and mechanical properties were studied. Streptozotocin induced diabetes, with significant worsening of bone mineralization and bone mechanical properties. Streptozotocin after nicotinamide induced slight glycemia increases in first days of experiment only, however worsening of cancellous bone mechanical properties and decreased vertebral bone mineral density (BMD) were demonstrated. Trigonelline decreased bone mineralization and tended to worsen bone mechanical properties in streptozotocin-induced diabetic rats. In nicotinamide/streptozotocin-treated rats, Trigonelline significantly increased BMD and tended to improve cancellous bone strength. Trigonelline differentially affected the skeletal system of rats with streptozotocin-induced metabolic disorders, intensifying the osteoporotic changes in streptozotocin-treated rats and favorably affecting bones in the non-hyperglycemic (nicotinamide/streptozotocin-treated) rats. The results indicate that, in certain conditions, Trigonelline may damage bone.
Keywords: coffee; diabetes; rats; skeletal system; streptozotocin; Trigonelline. | | Nutrients . 2016 Mar 2;8(3):133. | | Effects of Trigonelline, an Alkaloid Present in Coffee, on Diabetes-Induced Disorders in the Rat Skeletal System[Pubmed: 26950142] | | Abstract
Diabetes increases bone fracture risk. Trigonelline, an alkaloid with potential antidiabetic activity, is present in considerable amounts in coffee. The aim of the study was to investigate the effects of Trigonelline on experimental diabetes-induced disorders in the rat skeletal system. Effects of Trigonelline (50 mg/kg p.o. daily for four weeks) were investigated in three-month-old female Wistar rats, which, two weeks before the start of Trigonelline administration, received streptozotocin (60 mg/kg i.p.) or streptozotocin after nicotinamide (230 mg/kg i.p.). Serum bone turnover markers, bone mineralization, and mechanical properties were studied. Streptozotocin induced diabetes, with significant worsening of bone mineralization and bone mechanical properties. Streptozotocin after nicotinamide induced slight glycemia increases in first days of experiment only, however worsening of cancellous bone mechanical properties and decreased vertebral bone mineral density (BMD) were demonstrated. Trigonelline decreased bone mineralization and tended to worsen bone mechanical properties in streptozotocin-induced diabetic rats. In nicotinamide/streptozotocin-treated rats, Trigonelline significantly increased BMD and tended to improve cancellous bone strength. Trigonelline differentially affected the skeletal system of rats with streptozotocin-induced metabolic disorders, intensifying the osteoporotic changes in streptozotocin-treated rats and favorably affecting bones in the non-hyperglycemic (nicotinamide/streptozotocin-treated) rats. The results indicate that, in certain conditions, Trigonelline may damage bone.
Keywords: coffee; diabetes; rats; skeletal system; streptozotocin; Trigonelline. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)