| Structure Identification: |
| Appl Microbiol Biotechnol. 2013 Jun;97(12):5275-82. | | Regioselective synthesis of flavonoid bisglycosides using Escherichia coli harboring two glycosyltransferases.[Pubmed: 23549747 ] | Regioselective glycosylation of flavonoids cannot be easily achieved due to the presence of several hydroxyl groups in flavonoids.
METHODS AND RESULTS:
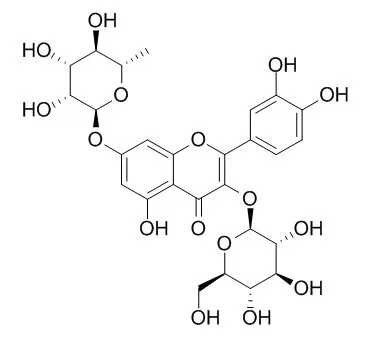
This hurdle could be overcome by employing uridine diphosphate-dependent glycosyltransferases (UGTs), which use nucleotide sugars as sugar donors and diverse compounds including flavonoids as sugar acceptors. Quercetin rhamnosides contain antiviral activity. Two quercetin diglycosides, Quercetin 3-O-glucoside-7-O-rhamnoside and quercetin 3,7-O-bisrhamnoside, were synthesized using Escherichia coli expressing two UGTs. For the synthesis of Quercetin 3-O-glucoside-7-O-rhamnoside, AtUGT78D2, which transfers glucose from UDP-glucose to the 3-hydroxyl group of quercetin, and AtUGT89C1, which transfers rhamnose from UDP-rhamnose to the 7-hydroxyl group of quercetin 3-O-glucoside, were transformed into E. coli. Using this approach, 67 mg/L of Quercetin 3-O-glucoside-7-O-rhamnoside was synthesized. For the synthesis of quercetin 3,7-O-bisrhamnoside, AtUGT78D1, which transfers rhamnose to the 3-hydroxy group of quercetin, and AtUGT89C1 were used.
CONCLUSIONS:
The RHM2 gene from Arabidopsis thaliana was coexpressed to supply the sugar donor, UDP-rhamnose. E. coli expressing AtUGT78D1, AtUGT89C1, and RHM2 was used to obtain 67.4 mg/L of quercetin 3,7-O-bisrhamnoside. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)