| Structure Identification: |
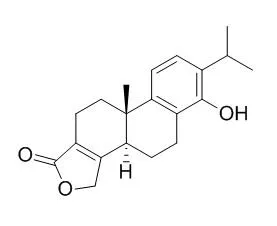
| Yao Xue Xue Bao. 1992;27(11):867-70. | | The crystal structure of triptophenolide revisited.[Pubmed: 1300033] | | Triptophenolide, C20H24O3, has been assigned two isomeric structures. DF Yu et al reported the X-ray analytical structure II in 1990. In 1982, however, FX Deng et al had already reported the spectrometric assignment of structure I. Now, we report again the single crystal analysis of Triptophenolide (I). The crystal belongs to space group P2(1)2(1)2(1) of orthorhombic system, with a = 7.270(6), b = 12.509(8), c = 36.20(1) A, Z = 8. The structure was solved by direct method and refined by the full-matrix least-square method to a final R factor 0.055, based on 2322 intensities with I > or = 3 sigma (I). It is highly probable that the result of DF Yu was in error with only 1292 available intensities. | | J Org Chem. 2000 Apr 7;65(7):2208-17. | | Enantioselective total synthesis of (-)-triptolide, (-)-triptonide, (+)-triptophenolide, and (+)-triptoquinonide.[Pubmed: 10774048] | | The first enantioselective total synthesis of (-)-triptolide (1), (-)-triptonide (2), (+)-Triptophenolide (3), and (+)-triptoquinonide (4) was completed. The key step involves lanthanide triflate-catalyzed oxidative radical cyclization of (+)-8-phenylmenthyl ester 30 mediated by Mn(OAc)3, providing intermediate 31 with good chemical yield (77%) and excellent diastereoselectivity (dr 38:1). (+)-Triptophenolide methyl ether (5) was then prepared in > 99% enantiomeric excess (> 99% ee), and readily converted to natural products 1-4. In addition, transition state models were proposed to explain the opposite chiral induction observed in the oxidative radical cyclization reactions of chiral beta-keto esters 17 (without an alpha-substituent) and 17a (with an alpha-chloro substituent). |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)