| Structure Identification: |
| Nat Prod Res. 2015 Feb;29(3):253-61. | | Isolation, structural elucidation and cytotoxicity evaluation of a new pentahydroxy-pimarane diterpenoid along with other chemical constituents from Aerva lanata.[Pubmed: 25348942] | Aervalanata possesses various useful medicinal and pharmaceutical activities.
METHODS AND RESULTS:
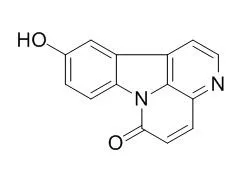
Phytochemical investigation of the plant has now led to the isolation of a new 2α,3α,15,16,19-pentahydroxy pimar-8(14)-ene diterpenoid (1) together with 12 other known compounds identified as β-sitosterol (2), β-sitosterol-3-O-β-D-glucoside (3), canthin-6-one (4), 10-Hydroxycanthin-6-one (aervine, 5), 10-methoxycanthin-6-one (methylaervine, 6), β-carboline-1-propionic acid (7), 1-O-β-D-glucopyranosyl-(2S,3R,8E)-2-[(2'R)-2-hydroxylpalmitoylamino]-8-octadecene-1,3-diol (8), 1-O-(β-D-glucopyranosyl)-(2S,3S,4R,8Z)-2-[(2'R)-2'-hydroxytetracosanoylamino]-8(Z)-octadene-1,3,4-triol (9), (2S,3S,4R,10E)-2-[(2'R)-2'-hydroxytetracosanoylamino]-10-octadecene-1,3,4-triol (10), 6'-O-(4″-hydroxy-trans-cinnamoyl)-kaempferol-3-O-β-D-glucopyranoside (tribuloside, 11), 3-cinnamoyltribuloside (12) and sulfonoquinovosyldiacylglyceride (13). Among these, six compounds (8-13) are reported for the first time from this plant.
CONCLUSIONS:
Cytotoxicity evaluation of the compounds against five cancer cell lines (CHO, HepG2, HeLa, A-431 and MCF-7) shows promising IC50 values for compounds 4, 6 and 12. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)