| In vitro: |
| Biosci Biotechnol Biochem. 2015 May;79(5):700-6. | | Tricin derivatives as anti-inflammatory and anti-allergic constituents from the aerial part of Zizania latifolia.[Pubmed: 25559019] |
METHODS AND RESULTS:
Methanol extract of Zizania latifolia was partitioned with EtOAc, n-BuOH, and H2O. From the EtOAc layers, a new flavonolignan along with a known flavone and three known flavonolignans, Tricin (1), salcolin A (2), salcolin B (3), and salcolin C (4), were isolated through repeated silica gel and ODS column chromatography. The chemical structure of the new flavonolignan was determined to be Tricin-4'-O-[erythro-β-guaiacyl-(7″-O-methyl)-glyceryl] ether and was named salcolin D (5) based on physicochemical and spectroscopic data, including FT-NMR and ESI-MS. All compounds were isolated for the first time from this plant. Compounds 2-5, Tricin derivatives, all exhibited higher anti-inflammatory and anti-allergy activities than Tricin. In particular, salcolin D (5) was shown to have the strongest inhibitory activity against LPS-induced NO production in RAW 264.7 cells as well as β-hexosaminidase release in IgE-sensitized RBL-2H3 cells.
CONCLUSIONS:
These results suggest that the presence of Tricin derivatives conveys allergy and inflammation treatment ability to Z. latifolia. | | Microbes Infect. 2012 Oct;14(12):1086-92. | | Anti-cytomegalovirus effects of tricin are dependent on CXCL11.[Pubmed: 22683667] | It has been reported that treatment with Tricin (4',5,7-trihydroxy-3',5'-dimethoxyflavone), a derivative of Sasa albo-marginata, after human cytomegalovirus (HCMV) infection significantly suppressed both infectious virus production and HCMV replication in the human embryonic fibroblast cell line MRC-5.
METHODS AND RESULTS:
In this paper, we examined the mechanisms for the anti-HCMV effects of Tricin in MRC-5 cells. Exposure of fibroblasts to Tricin inhibited infectious HCMV production, with concomitant decreases in levels of transcripts of the CXC chemokine IFN-inducible T cell alpha chemoattractant (I-TAC or CXCL11) gene. We also found that the transcripts of the HCMV immediate early (IE) gene and replication of HCMV were lower in CXCL11 gene-knockdown cells.
CONCLUSIONS:
These results suggest that Tricin is a novel compound with potential anti-HCMV activity and that CXCL11 is one of the chemokines involved in HCMV replication. In addition, it is possible that CXCL11 is the one of the targets of Tricin. | | J Agric Food Chem . 2018 Jul 5;66(26):6708-6716. | | Inhibition of the Proliferation and Invasion of C6 Glioma Cells by Tricin via the Upregulation of Focal-Adhesion-Kinase-Targeting MicroRNA-7[Pubmed: 29877083] | | Abstract
Tricin, a natural flavonoid present in large amounts in rice bran, was investigated for the mechanisms by which it exhibited antiproliferation and anti-invasion in C6 glioma cells. The results indicated that treatment with 5, 10, 25, and 50 μM Tricin for 48 h significantly ( p < 0.05) inhibited cell numbers and colony numbers with values of 134.3 ± 5.5, 114.6 ± 2.5, 106.3 ± 3.2, and 57.3 ± 10.2, respectively. Tricin also inhibited C6-cell motility, migration, and invasion. Tricin changed the cytoskeletal organization, reduced matrix-metalloproteinase (MMP) expression, and upregulated E-cadherin. Tricin decreased FAK protein levels and suppressed focal-adhesion-kinase (FAK)-downstream-signal activation. Most importantly, Tricin dose-dependently upregulated microRNA-7 (miR-7). Transfection with an miR-7 inhibitor suppressed miR-7 expression, increased FAK expression, and promoted the proliferation and invasion in C6 cells. The data support a novel anticancer mechanism of Tricin that involves upregulation of FAK-targeting miR-7 in C6 glioma cells.
Keywords: anticancer; focal-adhesion kinase; glioma; microRNA-7; Tricin. |
|


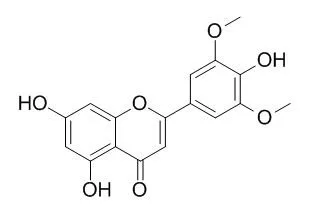



 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)