| Structure Identification: |
| Yao Xue Xue Bao. 1992;27(10):748-51. | | Studies on the chemical constituents of Glycyrrhiza pallidiflora Maxim.[Pubmed: 1293921] | A species of the genus Glycyrrhiza, G. pallidiflora Maxim, growing in Jiangsu and Hebei Provices of China, has been little studied before on its chemical constituents. This paper reports the isolation and chemical elucidation of five compounds from this plant, one of them is a new compound named glypallichalcone (P-2).
METHODS AND RESULTS:
Their chemical structures were elucidated by means of chemical and spectrometric analysis (UV, IR, MS, 1HNMR and 13CNMR) and were first reported to be present in this plant. Glypallichalcone (P-2), was found to be 4-hydroxy-2,4'-dimethoxy-chalcone.
CONCLUSIONS:
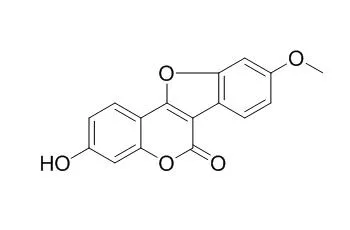
The known compounds were identified to be 4'-O-Methylcoumestrol (P-1), N-acetyl-glutamic acid (P-3), formononetin and beta-sitosterol (P-5). | | J Org Chem. 2017 Oct 20;82(20):11102-11109. | | Collective Syntheses of 2-(3-Methylbenzofuran-2-yl)phenol-Derived Natural Products by a Cascade [3,3]-Sigmatropic Rearrangement/Aromatization Strategy.[Pubmed: 28944672] |
METHODS AND RESULTS:
A cascade [3,3]-sigmatropic rearrangement/aromatization strategy to the synthesis of 2-(3-methylbenzofuran-2-yl)phenol derivatives was developed and applied to the collective syntheses of seven 2-arylbenzofuran-containing natural products, namely glycybenzofuran, glycyuralin E, lespedezol A1, puerariafuran, 7,2',4'-trihydroxy-3-benzofurancarboxylic acid, coumestrol, and 4'-O-Methylcoumestrol. Among them, the total syntheses of glycybenzofuran, glycyuralin E, puerariafuran, 7,2',4'-trihydroxy-3-benzofurancarboxylic acid, and 4'-O-Methylcoumestrol were reported for the first time.
CONCLUSIONS:
The practicality of this novel strategy in preparation of the key intermediates was demonstrated by performing the reaction on gram scale and by synthesizing a series of natural products with 2-(3-methylbenzofuran-2-yl)phenol scaffolds in a common strategy. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)