| In vitro: |
| Biological Sciences, 2007. | | Studies on Diterpenoid Constituents from Taxus sumatrana in Taiwan.[Reference: WebLink] | Taxol is a complex polyoxygenated diterpene isolated from Pacific yew (Taxus brevifolia). The structures of Taxoids are diversified with species, season and growth environment and the clinical effectiveness of Taxol as a microtubule-stabilizing therapeutic agent for treatment of several malignancies has motivated many scientists to isolate new taxoids and investigate their anti-tumor activities.
METHODS AND RESULTS:
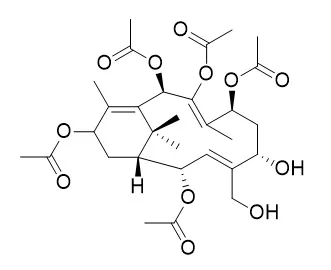
In this continuing search for new and bioactive natural taxoids, reinvestigation of the acetone extract of the twigs, needles and branches of Taxus sumatrana (Taxaceae) afforded thirty-seven taxane diterpenes esters, including sumataxins sumataxin A (1)¡Bsumataxin B (2)¡Bsumataxin C (3)¡Bsumataxin D (4)¡Btaxuyunnanine C (5)¡B5a,7B,9a,10B,13a-petaacetoxy-4(20),11-taxadiene (6)¡B2a,5a,9a,10B,14B-pentaacetoxytaxa-4(20),11-taxadiene (7)¡B14B-hydroxytaxusin (8)¡B2a-deacetoxytaxinine J (9)¡Btaxa-4-(20),11-diene-2a,5a,7B,9a,10B,13a-hexaol hexaacetate (10)¡B1-dehydroxy baccatin VI (11)¡B7B,9a,10B,13a,20-pentaacetoxy-2a-benzoyloxy-4a,5a-dihydroxytax-11-ene (12)¡Btaxacin (13)¡Bbaccatin VI (14)¡Btaxuspinanane J (15)¡B2-deacetoxy-5-decinnamoyltaxinine J (16)¡BN-Methyl taxol C (17)¡B10-deacetyl yunnanaxane (18)¡Btaxumairol B (19)¡Btaxinine M (20)¡Bbaccatin III (21)¡Btaxuspinanane I (22)¡Btaxumairol K (23)¡Bwallifoliol (24)¡B13-oxo-baccatin III (25)¡Btaxol (26)¡B7-epi-10-deacetyl taxol (27)¡B10-deacetyl-13-oxo-baccatin III (28)¡B19-hydroxybaccatin III (29)¡B10-deacetyl taxol (30)¡B10-deacetyl-baccatin III (31)¡B13-acetyl-13-decinamoyltaxachinin B (32)¡B5-deacetyltaxachitriene B (33)¡B5-Epicanadensene (34)¡Btaxezopidine F (35)¡B13a,7B-diacetoxy-2a,5a,10B-trihydroxy-9-keto-2(3¡÷20)abeotaxane (36)¡B2-deacetyl taxine B (37). The structures of new compounds were established on the basis of their spectroscopic analyses.
CONCLUSIONS:
Among them, compounds 1, 2, 3 and 4 are new compounds from natural source, 2a-deacetoxytaxinine J (9)¡Btaxuspinanane J (15) had effects on PBMC (Peripheral Blood Mononuclear Cells) proliferation, and sumataxin A¡BD (1¡B4) had light exhibitedactivity of HSV-1. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)