| Description: |
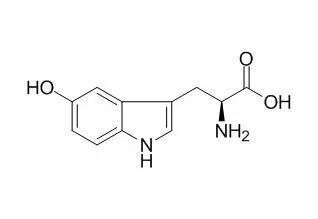
5-Hydroxytryptophan (5-HTP) is the precursor of serotonin , has been widely used as a dietary supplement to raise serotonin level, it is an effective drug against depression, insomnia, obesity, chronic headaches, etc. 5-HTP supplementation can inhibit endothelial serotonylation, leukocyte recruitment, and allergic inflammation, it also can reduce the symptoms of anxiety/depression of that patients with allergy/asthma. 5-HTP
may be involved in inducing the female to stay in copula and delay egg-laying to prevent re-mating of the female. |
| Targets: |
NF-kB | IL Receptor | TNF-α |
| In vitro: |
| Am J Physiol Lung Cell Mol Physiol. 2012 Oct 15; 303(8): L642–L660. | | Inhibition of allergic inflammation by supplementation with 5-hydroxytryptophan[Pubmed: 22842218] | Clinical reports indicate that patients with allergy/asthma commonly have associated symptoms of anxiety/depression. Anxiety/depression can be reduced by 5-Hydroxytryptophan (5-HTP) supplementation. However, it is not known whether 5-HTP reduces allergic inflammation. Therefore, we determined whether 5-HTP supplementation reduces allergic inflammation. We also determined whether 5-HTP decreases passage of leukocytes through the endothelial barrier by regulating endothelial cell function. For these studies, C57BL/6 mice were supplemented with 5-HTP, treated with ovalbumin fraction V (OVA), house dust mite (HDM) extract, or IL-4, and examined for allergic lung inflammation and OVA-induced airway responsiveness.
METHODS AND RESULTS:
To determine whether 5-HTP reduces leukocyte or eosinophil transendothelial migration, endothelial cells were pretreated with 5-HTP, washed and then used in an in vitro transendothelial migration assay under laminar flow. Interestingly, 5-HTP reduced allergic lung inflammation by 70–90% and reduced antigen-induced airway responsiveness without affecting body weight, blood eosinophils, cytokines, or chemokines. 5-HTP reduced allergen-induced transglutaminase 2 (TG2) expression and serotonylation (serotonin conjugation to proteins) in lung endothelial cells. Consistent with the regulation of endothelial serotonylation in vivo, in vitro pretreatment of endothelial cells with 5-HTP reduced TNF-α-induced endothelial cell serotonylation and reduced leukocyte transendothelial migration. Furthermore, eosinophil and leukocyte transendothelial migration was reduced by inhibitors of transglutaminase and by inhibition of endothelial cell serotonin synthesis, suggesting that endothelial cell serotonylation is key for leukocyte transendothelial migration.
CONCLUSIONS:
In summary, 5-HTP supplementation inhibits endothelial serotonylation, leukocyte recruitment, and allergic inflammation. These data identify novel potential targets for intervention in allergy/asthma. |
|
| In vivo: |
| Arch Oral Biol. 2015 May;60(5):789-98. | | Oral administration of 5-hydroxytryptophan aggravated periodontitis-induced alveolar bone loss in rats.[Pubmed: 25766472] | 5-Hydroxytryptophan (5-HTP) is the precursor of serotonin and 5-Hydroxytryptophan has been widely used as a dietary supplement to raise serotonin level. Serotonin has recently been discovered to be a novel and important player in bone metabolism. As peripheral serotonin negatively regulates bone, the regular take of 5-Hydroxytryptophan may affect the alveolar bone metabolism and therefore influence the alveolar bone loss induced by periodontitis. The aim of this study was to investigate the effect of 5-Hydroxytryptophan on alveolar bone destruction in periodontitis.
METHODS AND RESULTS:
Male Sprague-Dawley rats were randomly divided into the following four groups: (1) the control group (without ligature); (2) the 5-HTP group (5-Hydroxytryptophan at 25 mg/kg/day without ligature); (3) the L group (ligature+saline placebo); and (4) the L+5-Hydroxytryptophan group (ligature+5-Hydroxytryptophan at 25 mg/kg/day). Serum serotonin levels were determined by ELISA. The alveolar bones were evaluated with micro-computed tomography and histology. Tartrate-resistant acid phosphatase staining was used to assess osteoclastogenesis. The receptor activator of NF-kB ligand (RANKL) and osteoprotegerin (OPG) expression in the periodontium as well as the interleukin-6 positive osteocytes were analysed immunohistochemically. 5-Hydroxytryptophan significantly increased serum serotonin levels. In rats with experimental periodontitis, 5-Hydroxytryptophan increased alveolar bone resorption and worsened the micro-structural destruction of the alveolar bone. 5-Hydroxytryptophan also stimulated osteoclastogenesis and increased RANKL/OPG ratio and the number of IL-6 positive osteocytes. However, 5-Hydroxytryptophan treatment alone did not cause alveolar bone loss in healthy rats.
CONCLUSIONS:
The present study showed that 5-Hydroxytryptophan aggravated alveolar bone loss, deteriorated alveolar bone micro-structure in the presence of periodontitis, which suggests 5-Hydroxytryptophan administration may increase the severity of periodontitis. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)