| Description: |
7,8-Dihydroxyflavone(7,8-DHF) shows vasorelaxing , antihypertensive, anti-inflammatory and anti-oxidant properties, it can be used as a potent facultative ingredient in health-beneficial agents to prevent or treat the skin aging or inflammatory skin disorders. 7,8-DHF has anti-obesity activity, it improves mitochondrial respiration in STs from obese women, it is beneficial for both depression and anxiety-like behaviors, and may exert fast-onset antidepressant effects. 7,8-DHF exhibits anti-melanogenic activity through inhibition of tyrosinase activity in α-MSH-stimulating condition, it also shows anticancer activity in melanoma cells via downregulation of α-MSH/cAMP/MITF pathway. 7,8-DHF ameliorates motor deficits via suppressing α-synuclein expression and oxidative stress in the MPTP-induced mouse model of Parkinson's disease. |
| In vitro: |
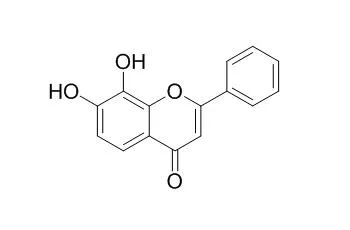
| Biomed Pharmacother. 2017 Sep 22;95:1580-1587. | | 7,8-Dihydroxyflavone attenuates TNF-α-induced skin aging in Hs68 human dermal fibroblast cells via down-regulation of the MAPKs/Akt signaling pathways.[Pubmed: 28950658 ] | 7,8-Dihydroxyflavone (7,8-DHF, 7,8-dihydroxy-2-phenyl-4H-chromen-4-one) is a natural flavone found in plants and has been frequently reported to show anti-inflammatory and anti-oxidant properties. Skin aging is induced mainly by oxidative stress.
METHODS AND RESULTS:
In the present study, we evaluated 7,8-DHF for its potential anti-aging effects for skin using Hs68 human dermal fibroblast cells.
To establish aged skin cell model, Hs68 cells were treated with tumor necrosis factor-α (TNF-α) for 18h 7,8-DHF (0-10μM) induced collagen synthesis and suppressed the expression of matrix metalloproteinase 1 (MMP 1) in a dose-dependent manner. 7,8-DHF also significantly reduced the generation of intracellular reactive oxygen species (ROS), induced the expression of anti-oxidant enzymes, such as catalase, manganese superoxide dismutase (Mn-SOD), and heme oxygenase-1 (HO-1), and scavenged DPPH free radicals. 7,8-DHF also disturbed the mitogen-activated protein kinases (MAPKs) and Akt signaling pathways that participate in the aging process. 7,8-DHF exerted potent anti-aging effects by inhibiting MMP 1 expression and inducing Type I collagen synthesis in Hs68 cells. 7,8-DHF effectively attenuated oxidative stress by up-regulating the anti-oxidant enzymes catalase, Mn-SOD, and HO-1, and reducing activation of the Akt and MAPKs signaling pathways in aged skin cells.
CONCLUSIONS:
These results suggest that 7,8-DHF can be used as a potent facultative ingredient in health-beneficial agents to prevent or treat the skin aging or inflammatory skin disorders. | | Life Sci. 2016 Jan 1;144:103-12. | | 7,8-Dihydroxyflavone inhibits adipocyte differentiation via antioxidant activity and induces apoptosis in 3T3-L1 preadipocyte cells.[Pubmed: 26631505 ] | Anti-obesity effects of a natural plant flavonoid 7,8-Dihydroxyflavone (7,8-DHF) were evaluated using 3T3-L1 preadipocyte cells.
METHODS AND RESULTS:
The cell viability was determined using MTT assay. Effects of 7,8-DHF on intracellular lipid droplets and intracellular reactive oxygen species (ROS) were measured using a 2,7-dichlorofluorescein diacetate (DCF-DA) assay and Oil Red O staining method, respectively. Apoptotic cell death was monitored by annexin V-FITC/PI double staining and by a TUNEL assay. Antioxidant enzyme mRNA levels and protein expression of adipogenic transcription factors were determined by real-time PCR and Western blotting, respectively.
Whereas the cell viability of 3T3-L1 preadipocytes was not affected by lower concentrations of 7,8-DHF (<20 μM), higher concentrations of 7,8-DHF (>20 μM) induced apoptotic cell death. 7,8-DHF (<20 μM) significantly reduced the intracellular lipid droplets and the expression of major adipogenic transcription factors, such as CCAAT/enhancer-binding protein-α (C/EBP-α), C/EBP-β, and peroxisome proliferator activated receptor-γ (PPAR-γ). 7,8-DHF treatment also dose-dependently reduced the intracellular ROS level, attenuated MAPK pathway activation, and increased the expression of antioxidant enzymes, such as Mn-superoxide dismutase (Mn-SOD), catalase (CAT), and heme oxygenase-1 (HO-1).
CONCLUSIONS:
The results of this study indicated that 7,8-DHF inhibits the adipogenesis of 3T3-L1 preadipocyte cells by down-regulating the expression of adipogenic transcription factors, reduces lipid accumulation, and attenuates ROS accumulation by inducing antioxidant enzymes in differentiated 3T3-L1 cells, suggesting for the first time that 7,8-DHF has an anti-obesity effect in vitro via its anti-oxidant activity. |
|
| In vivo: |
| Exp Neurol. 2017 Sep 4;298(Pt A):79-96. | | A flavonoid agonist of the TrkB receptor for BDNF improves hippocampal neurogenesis and hippocampus-dependent memory in the Ts65Dn mouse model of DS.[Pubmed: 28882412 ] | Intellectual disability is the unavoidable hallmark of Down syndrome (DS), with a heavy impact on public health. Reduced neurogenesis and impaired neuron maturation are considered major determinants of altered brain function in DS. Since the DS brain starts at a disadvantage, attempts to rescue neurogenesis and neuron maturation should take place as soon as possible.
The brain-derived neurotrophic factor (BDNF) is a neurotrophin that plays a key role in brain development by specifically binding to tropomyosin-related kinase receptor B (TrkB).
Systemic BDNF administration is impracticable because BDNF has a poor blood-brain barrier penetration. Recent screening of a chemical library has identified a flavone derivative, 7,8-Dihydroxyflavone (7,8-DHF), a small-molecule that crosses the blood-brain barrier and binds with high affinity and specificity to the TrkB receptor. The therapeutic potential of TrkB agonists for neurogenesis improvement in DS has never been examined.
METHODS AND RESULTS:
The goal of our study was to establish whether it is possible to restore brain development in the Ts65Dn mouse model of DS by targeting the TrkB receptor with 7,8-DHF. Ts65Dn mice subcutaneously injected with 7,8-DHF in the neonatal period P3-P15 exhibited a large increase in the number of neural precursor cells in the dentate gyrus and restoration of granule cell number, density of dendritic spines and levels of the presynaptic protein synaptophysin. In order to establish the functional outcome of treatment, mice were treated with 7,8-DHF from P3 to adolescence (P45-50) and were tested with the Morris Water Maze. Treated Ts65Dn mice exhibited improvement of learning and memory, indicating that the recovery of the hippocampal anatomy translated into a functional rescue.
CONCLUSIONS:
Our study in a mouse model of DS provides novel evidence that treatment with 7,8-DHF during the early postnatal period restores the major trisomy-linked neurodevelopmental defects, suggesting that therapy with 7,8-DHF may represent a possible breakthrough for Down syndrome. | | Reprod Sci. 2017 Jan 1:1933719117716776. | | Tropomyosin Receptor Kinase B Agonist, 7,8-Dihydroxyflavone, Improves Mitochondrial Respiration in Placentas From Obese Women.[Pubmed: 28677406] | Maternal obesity negatively impacts the placenta, being associated with increased inflammation, decreased mitochondrial respiration, decreased expression of brain-derived neurotrophic factor (BDNF), and its receptor, tropomyosin receptor kinase B (TRKB). TRKB induction by 7,8-Dihydroxyflavone (7,8-DHF) improves energy expenditure in an obesity animal model.
METHODS AND RESULTS:
We hypothesized that TRKB activation would improve mitochondrial respiration in trophoblasts from placentas of obese women. Placentas were collected from lean (pre-pregnancy BMI < 25) and obese (pre-pregnancy BMI > 30) women at term following cesarean section delivery without labor. Cytotrophoblasts were isolated and plated, permitting syncytialization. At 72 hours, syncytiotrophoblasts (STs) were treated for 1 hour with 7,8-DHF (10 nM-10 M), TRKB antagonists (ANA-12 (10 nM-1 M), Cyclotraxin B (1 nM-1M)), or vehicle. Mitochondrial respiration was measured using the XF24 Extracellular Flux Analyzer. TRKB, MAPK, and PGC1α were measured using Western blotting. Maternal obesity was associated with decreased mitochondrial respiration in STs; however, 7,8-DHF increased basal, ATP-coupled, maximal, spare capacity, and nonmitochondrial respiration. A 10 μM dose of 7,8-DHF reduced spare capacity in STs from lean women, with no effect on other respiration parameters. 7,8-DHF had no effect on TRKB phosphorylation; however, there was a concentration-dependent decrease of p38 MAPK phosphorylation and increase of PGC1α in STs from obese, but not in lean women. TRKB antagonism attenuated ATP-coupled respiration, maximal respiration, and spare capacity in STs from lean and obese women.
CONCLUSIONS:
7,8-DHF improves mitochondrial respiration in STs from obese women, suggesting that the obese phenotype in the placenta can be rescued by TRKB activation. | | Am J Hypertens. 2014 May;27(5):750-60. | | Vasorelaxing and antihypertensive effects of 7,8-dihydroxyflavone.[Pubmed: 24317273 ] | Although 7,8-Dihydroxyflavone (7,8-DHF) has been demonstrated to be potently neuroprotective, its effect on vascular function remains unknown.
METHODS AND RESULTS:
The effect of 7,8-DHF on phenylephrine (PE)-induced preconstriction was examined with aortic rings isolated from normal rats. Its effective mechanisms were studied with blockers, Western blotting, and primarily cultured vascular smooth myocytes. The blood pressure (BP) of rats was measured with a tail cuff method.
7,8-DHF dose-dependently dilated the PE-preconstricted, endothelia-intact aortic rings with concentration for 50% of maximal effect (EC50) of approximately 24 µM. Both Nω-nitro-L-arginine methyl ester hydrochloride, a nitric oxide synthase inhibitor, and 1H-[1,2,4]oxadiazolo[4,3-a]quinoxalin-1-one, a soluble guanylyl cyclase blocker, significantly reduced the vasorelaxing effect of 7,8-DHF. Western blotting showed that 7,8-DHF increased the aortic endothelial nitric oxide synthase protein expression and phosphorylation. With endothelia removed, 7,8-DHF also dilated the PE-preconstricted rings but with EC50 of approximately 104 µM. Ca(2+) imaging experiments detected that 7,8-DHF probably blocked both intracellular Ca(2+) release and extracellular Ca(2+) influx. Therefore, the mechanisms of 7,8-DHF dilating effect might be stimulating the nitric oxide/cGMP production and blocking the Ca(2+) signaling pathway instead of tropomyosin receptor kinase B receptors because ANA-12, its specific antagonist, did not show any effect against 7,8-DHF. When administered intravenously, 7,8-DHF significantly reduced the BP of the spontaneously hypertensive rats. However, when used orally, there was only a slight but significant reduction in the diastolic pressure.
CONCLUSIONS:
The results suggest that neuro-protective 7,8-DHF is also a vasorelaxing and antihypertensive substance in rats. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)