| Description: |
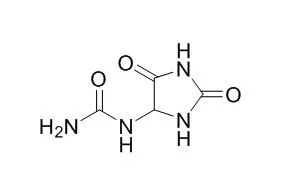
Allantoin, as I-1R agonist, has the potential to develop as a new therapeutic agent for hypertension. Allantoin has memory-enhancing, anti-oxidative and anti-inflammatory activities; it can enhance the antifungal activity of Nanoencapsulation. Allantoin is a skin conditioning agent that promotes healthy skin, stimulates new and healthy tissue growth. Allantoin mediates PI3K-Akt-GSK-3β signal pathway.
|
| Targets: |
PI3K | Akt | GSK-3 | Antifection | Imidazoline I-1 receptor |
| In vitro: |
| Pharmazie. 2015 Mar;70(3):155-64. | | Solid lipid nanoparticles containing copaiba oil and allantoin: development and role of nanoencapsulation on the antifungal activity.[Pubmed: 25980176] | The aim of this work was to develop solid lipid nanoparticles (SLN) containing copaiba oil with and without Allantoin (NCOA, NCO, respectively) and to evaluate their antifungal activity.
METHODS AND RESULTS:
The dynamic light scattering analysis showed z-average diameters (intensity) between 118.63 ± 8.89 nm for the nanoparticles with both copaiba oil and Allantoin and 126.06 ± 9.84nm for the nanoparticles with just copaiba oil. The D[4,3] determined by laser diffraction showed similar results of 123 ± 1.73 nm for the nanoparticles with copaiba oil and Allantoin and 130 ± 3.6 nm for the nanoparticles with copaiba oil alone. Nanoparticle tracking analysis demonstrated that both suspensions had monomodal profiles and consequently, the nanoparticle populations were homogeneous. This analysis also corroborated the results of dynamic light scattering and laser diffraction, exhibiting a smaller mean diameter for the nanoparticles with copaiba oil and Allantoin (143 nm) than for the nanoparticles with copaiba oil (204 nm). The nanoparticles with copaiba oil and the nanoparticles with copaiba oil and Allantoin presented a MIC90 of 500 μg/mL and 250 μg/mL, respectively, against C. krusei. The MIC90 values were 500 μg/mL (nanoparticles with copaiba oil) and 1.95 μg/mL (nanoparticles with copaiba oil and Allantoin) against T. rubrum. Against M. canis, the nanoparticles with copaiba oil and Allantoin had a MIC9 of 1.95 μg/mL.
CONCLUSIONS:
In conclusion, nanoencapsulation improved the antifungal activity of copaiba oil, which was enhanced by the presence of Allantoin. The MICs obtained are comparable to those of commercial products and can represent promising therapeutics for cutaneous infections caused by yeasts and dermatophytes. |
|
| In vivo: |
| Food Chem Toxicol. 2014 Feb;64:210-6. | | Effects of allantoin on cognitive function and hippocampal neurogenesis.[Pubmed: 24296131] | Allantoin is contained in Nelumbo nucifera (lotus) and a well-known cosmetic ingredient reported to have anti-oxidative and anti-inflammatory activities. In the present study, we investigated whether Allantoin affects cognitive function in mice.
METHODS AND RESULTS:
The subchronic administration of Allantoin (1, 3 or 10 mg/kg, for 7 days) significantly increased the latency time measured during the passive avoidance task in scopolamine-induced cholinergic blockade and normal naïve mice. Allantoin treatment (3 or 10 mg/kg, for 7 days) also increased the expression levels of phosphorylated phosphatidylinositide 3-kinase (PI3K), phosphorylated protein kinase B (Akt) and phosphorylated glycogen synthase kinase-3β (GSK-3β). Doublecortin and 5-bromo-2-deoxyuridine immunostaining revealed that Allantoin significantly increased the neuronal cell proliferation of immature neurons in the hippocampal dentate gyrus region. In conclusion, Allantoin has memory-enhancing effects, and these effects may be partly mediated by the PI3K-Akt-GSK-3β signal pathway.
METHODS AND RESULTS:
These findings suggest that Allantoin has therapeutic potential for the cognitive dysfunctions observed in Alzheimer's disease. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)